Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Qingdao Richkem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.richkem.com | |||
 | +86 (532) 8582-5696 | |||
 | +86 (532) 8582-5696 | |||
 | sales@richkem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shanghai Hongbang Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hongbangpharma.com | |||
 | +86 (21) 2428-0809 +86 13671516988 | |||
 | +86 (21) 2428-0809 | |||
 | kimberly-hongbangpharma@hotmail.com 1377976036@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Atul Potnis | India | |||
|---|---|---|---|---|
 | www.indoaminesltd.com | |||
 | +91 (251) 287-0941 287-1354 | |||
 | +91 (251) 287-1635 | |||
 | exports@indoaminesltd.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Kessie Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kessiechem.com | |||
 | +86 (519) 8698-7916 +86 15851999766 | |||
 | +86 (519) 8690-8212 | |||
 | info@kessiechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Changzhou Yuexing Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gmpharm.com | |||
 | +86 (519) 8506-6518 | |||
 | +86 (519) 8506-6519 | |||
 | quguosheng@vip.sina.com | |||
| Chemical manufacturer since 1989 | ||||
| Suzhou Jingye Medicine & Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.daxinchem.com | |||
 | +86 (512) 6665-8588 6665-8898 | |||
 | +86 (512) 6665-8801 | |||
 | info@jingyechem.cn | |||
| Chemical manufacturer since 1996 | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Dynamic International Enterprises Limited | China | |||
|---|---|---|---|---|
 | www.dynai.com | |||
 | +86 (21) 6231-2607 (510) 8755-1178 8755-4400 | |||
 | +86 (21) 6231-3801 / (510) 8755-1428 | |||
 | china@dynai.com | |||
| Chemical manufacturer | ||||
| Jintan Huayang Chemical Plant | China | |||
|---|---|---|---|---|
 | www.huayangchem.com | |||
 | +86 (519) 8252-1156 8252-7838 | |||
 | +86 (519) 8252-1118 | |||
 | hychem@huayangchem.com | |||
| Chemical manufacturer since 1992 | ||||
| Hubei Organic And Phosphoric Chemicals Import & Export Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hubeichem.com | |||
 | +86 (27) 8579-6109 8577-2811 | |||
 | +86 (27) 8577-3858 | |||
 | sales@hubeichem.com | |||
| Chemical distributor | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Liyang Yutian Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yutianchem.com | |||
 | +86 (519) 8719-0569 | |||
 | +86 (519) 8719-0111 | |||
 | yhh@yutianchem.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Jiangsu Tianyin Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinatianyin.com | |||
 | +86 (510) 8755-1178 | |||
 | +86 (510) 8755-1428 | |||
 | tianyin@chinatianyin.com | |||
| Chemical manufacturer | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Classification | Chemical pesticide >> Insecticide intermediate |
|---|---|
| Name | Benzylamine |
| Synonyms | alpha-Aminotoluene; Benzenemethanamine; Phenylmethylamine |
| Molecular Structure | 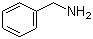 |
| Molecular Formula | C7H9N |
| Molecular Weight | 107.15 |
| CAS Registry Number | 100-46-9 |
| EC Number | 202-854-1 |
| SMILES | C1=CC=C(C=C1)CN |
| Density | 1.0±0.1 g/cm3 Calc.*, 0.981 g/mL (Expl.) |
|---|---|
| Melting point | 10 °C (Expl.) |
| Boiling point | 185.0 °C 760 mmHg (Calc.)*, 184 - 185 °C (Expl.) |
| Flash point | 60.0 °C (Calc.)*, 65 °C (Expl.) |
| Solubility | soluble (water) (Expl.) |
| Index of refraction | 1.547 (Calc.)*, 1.543 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H314-H318 Details | ||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P270-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 2735 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Benzylamine is an organic compound with the molecular formula C6H5CH2NH2. It is a primary amine consisting of a benzyl group (C6H5CH2–) attached to an amino group (–NH2). It appears as a colorless to slightly yellow liquid with a characteristic amine odor and is miscible with water and many organic solvents. Benzylamine is both a valuable intermediate in organic synthesis and an important reagent in research and industry. The compound was first characterized in the 19th century during the expansion of amine chemistry. Early studies focused on the reactivity of substituted amines and their preparation from corresponding halides or nitriles. Benzylamine could be synthesized by the reaction of benzyl chloride with ammonia or by the reduction of benzonitrile using catalytic hydrogenation. These classical methods remain useful, with refinements introduced to improve yield, selectivity, and safety. Benzylamine serves as a versatile building block in the preparation of a wide range of organic compounds. It participates in nucleophilic substitution reactions, condensation with aldehydes and ketones, acylation, and reductive amination processes. Its primary amine functionality allows it to form imines, amides, and other derivatives under mild conditions. Because of the relative ease of modifying both the benzyl and amine portions, it is widely employed in the synthesis of pharmaceuticals, agrochemicals, dyes, and polymers. In pharmaceutical chemistry, benzylamine is frequently used in the synthesis of active pharmaceutical ingredients (APIs) or as a precursor to substituted amines and heterocycles. Many biologically active compounds, including central nervous system agents, antihistamines, and cardiovascular drugs, feature benzylamine derivatives. Its presence contributes to both pharmacodynamic activity and physicochemical properties such as solubility and membrane permeability. In polymer and materials science, benzylamine is employed as a curing agent or modifier in the production of epoxy resins and polyurethanes. Its amine functionality reacts with epoxides to form cross-linked networks, which impart mechanical strength and chemical resistance to coatings and adhesives. Additionally, benzylamine can act as a surface modifier for silica, metal oxides, and carbon-based materials, enabling functionalization and dispersion in various matrices. In biochemical research, benzylamine has been studied as a substrate for the enzyme monoamine oxidase (MAO), particularly MAO-B. Its metabolism leads to the formation of benzaldehyde and hydrogen peroxide, making it a model compound for investigating amine oxidation mechanisms. Derivatives of benzylamine have also been explored as enzyme inhibitors and probes for receptor-binding studies. Benzylamine is compatible with a wide range of synthetic transformations, including reductive alkylation and amidation. It is stable under ambient conditions but should be stored under inert atmosphere to prevent oxidation or discoloration. Due to its volatility and reactive amine group, appropriate handling precautions are required, including avoidance of skin and eye contact and ventilation during use. Industrially, benzylamine is produced at scale for use as an intermediate in manufacturing dyes, optical brighteners, and chemical reagents. It is also used in the production of quaternary ammonium compounds, which serve as surfactants, disinfectants, and phase-transfer catalysts. Its reactivity with electrophiles and ability to form stable salts with acids make it suitable for diverse formulation needs. In summary, benzylamine is a valuable and well-characterized compound with broad utility in chemical synthesis, pharmaceutical development, materials science, and biochemical research. Its simple structure, high reactivity, and commercial availability make it an essential tool in both laboratory and industrial settings. References 2007. Alteration of serum semicarbazide-sensitive amine oxidase activity in chronic renal failure. Journal of neural transmission (Vienna, Austria : 1996), 114, 6. DOI: 10.1007/s00702-007-0698-4 2007. Studies on the insulinomimetic effects of benzylamine, exogenous substrate of semicarbazide-sensitive amine oxidase enzyme in streptozotocin induced diabetic rats. Journal of neural transmission (Vienna, Austria : 1996), 114, 6. DOI: 10.1007/s00702-007-0700-1 2007. Metabolism, Pharmacokinetics, and Excretion of a Nonpeptidic Substance P Receptor Antagonist, Ezlopitant, in Normal Healthy Male Volunteers: Characterization of Polar Metabolites by Chemical Derivatization with Dansyl Chloride. Drug metabolism and disposition: the biological fate of chemicals, 35, 7. DOI: 10.1124/dmd.107.015362 |
| Market Analysis Reports |