Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Feixiang Chemicals (Zhangjiagang) Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.feixiangchem.com | |||
 | +86 (512) 5811-0111 | |||
 | +86 (512) 5849-7000 | |||
 | sales@feixiangchem.com | |||
| Chemical manufacturer | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate |
|---|---|
| Name | N-Ethylmorpholine |
| Molecular Structure | 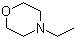 |
| Molecular Formula | C6H13NO |
| Molecular Weight | 115.17 |
| CAS Registry Number | 100-74-3 |
| EC Number | 202-885-0 |
| SMILES | CCN1CCOCC1 |
| Density | 0.905 |
|---|---|
| Melting point | -63 °C |
| Boiling point | 139 °C |
| Refractive index | 1.44-1.442 |
| Flash point | 27 °C |
| Water solubility | miscible |
| Hazard Symbols |     GHS02;GHS05;GHS06;GHS07 Danger Details GHS02;GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H302+H312+H332-H302-H311-H312-H314-H315-H318-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P262-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P319-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P363-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2920 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
N-Ethylmorpholine, a tertiary amine with the chemical formula C₆H₁₃NO, is an important organic compound in various industrial applications. This compound, which consists of a morpholine ring substituted by an ethyl group, is recognized for its utility as a catalyst, solvent, and intermediate in organic synthesis. The development and exploration of N-ethylmorpholine have contributed significantly to advancements in chemical manufacturing, particularly in the fields of pharmaceuticals and polymers. The discovery and initial use of N-ethylmorpholine date back to the mid-20th century when it was first synthesized by reacting morpholine with ethyl iodide. This straightforward alkylation reaction introduced the ethyl group into the morpholine structure, creating a versatile chemical that rapidly found applications in various chemical processes. The ease of its synthesis and the ability to modify its structure have made N-ethylmorpholine a valuable compound in both academic research and industrial applications. One of the primary applications of N-ethylmorpholine is as a catalyst in the production of polyurethane foams. Polyurethanes, widely used in furniture, insulation, and automotive industries, require catalysts that can promote the reaction between polyols and isocyanates. N-ethylmorpholine is particularly effective in this role, as it enhances the reaction rate while maintaining control over the foam’s cell structure and overall quality. The compound’s volatility also ensures that it does not remain in the final product, thus contributing to the production of foams with desirable physical properties. In addition to its role as a catalyst, N-ethylmorpholine serves as a solvent in various organic reactions. Its ability to dissolve both polar and nonpolar substances makes it suitable for use in a wide range of chemical processes, including the synthesis of pharmaceuticals and fine chemicals. N-ethylmorpholine is especially useful in reactions that require a solvent with moderate basicity, as it can facilitate the formation of desired products while minimizing side reactions. This has led to its widespread adoption in the pharmaceutical industry, where it is used in the manufacture of active pharmaceutical ingredients (APIs) and other drug intermediates. N-Ethylmorpholine is also employed as an intermediate in the synthesis of various organic compounds. Its reactivity, particularly its ability to undergo nucleophilic substitution and addition reactions, allows it to be used as a building block for more complex molecules. This versatility makes it a valuable starting material in the production of agrochemicals, dyes, and other specialty chemicals. Despite its usefulness, N-ethylmorpholine must be handled with care due to its potential health hazards. It is classified as an irritant, and exposure to the compound can cause skin, eye, and respiratory irritation. Proper safety precautions, including the use of personal protective equipment and adequate ventilation, are essential when working with N-ethylmorpholine. In summary, N-ethylmorpholine is a significant chemical compound with diverse applications in catalysis, solvent systems, and organic synthesis. Its discovery has enabled numerous advancements in the production of polyurethanes, pharmaceuticals, and other chemical products. As research and industrial processes continue to evolve, N-ethylmorpholine will likely remain a crucial component in various chemical applications, contributing to the ongoing development of new materials and technologies. References 1970. Determination of tertiary amines after formylation of primary and secondary amines with acetic-formic mixed anhydride. Fresenius' Zeitschrift fur analytische Chemie, 252(3). DOI: 10.1007/bf00424792 2021. Transition metal-based metal-organic frameworks for environmental applications: a review. Environmental Chemistry Letters, 19(3). DOI: 10.1007/s10311-020-01119-1 2022. The instability of a stable metal-organic framework in amino acid solutions. Nano Research, 15(8). DOI: 10.1007/s12274-022-4346-y |
| Market Analysis Reports |