Online Database of Chemicals from Around the World
| Nile Chemicals | India | |||
|---|---|---|---|---|
 | www.nilechemicals.com | |||
 | +91 (22) 6631-3162 | |||
 | +91 (22) 2205-4833 | |||
 | sales@nilechemicals.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Changsha Yonta Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hunanchem.com | |||
 | +86 (731) 8535-2228 | |||
 | +86 (731) 8535-2208 | |||
 | yontachem@gmail.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Changsha Hecom Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.emdchem.com | |||
 | +86 (731) 8578-1688 | |||
 | tan@emdchem.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shanxi Wencheng Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wenchengchem.com | |||
 | +86 (351) 799-1193 | |||
 | wenchengchem01@163.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chongqing Newcent New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinanewcent.com | |||
 | +86 (23) 8678-5320 | |||
 | sales2@chinanewcent.com | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Vishnupriya Chemicals Private Limited | India | |||
|---|---|---|---|---|
 | vishnupriya.in | |||
 | +91 9676770180 | |||
 | web@vishnupriya.in | |||
| Chemical manufacturer since 1979 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Cambridge Isotope Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.isotope.com | |||
 | +1 (978) 749-8000 | |||
 | +1 (978) 749-2768 | |||
 | cilsales@isotope.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| World Metal, LLC | USA | |||
|---|---|---|---|---|
 | www.worldmetalllc.com | |||
 | +1 (877) 363-8257 (281) 491-7474 | |||
 | +1 (281) 491-7979 | |||
 | sales@worldmetalllc.com | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Celtic Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.celticchemicals.co.uk | |||
 | +44 (1656) 749-358 | |||
 | +44 (1656) 746-490 | |||
 | sales@celticchemicals.co.uk | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal nitrates and nitrites |
|---|---|
| Name | Barium nitrate |
| Synonyms | barium(2+) dinitrate |
| Molecular Structure | 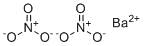 |
| Molecular Formula | Ba.(NO3)2 |
| Molecular Weight | 261.34 |
| CAS Registry Number | 10022-31-8 |
| EC Number | 233-020-5 |
| SMILES | [N+](=O)([O-])[O-].[N+](=O)([O-])[O-].[Ba+2] |
| Density | 3.23 g/mL (23 °C) (Expl.) |
|---|---|
| Melting point | 592 °C (Expl.) |
| Water solubility | 9 g/100 mL (20 °C) (Expl.) |
| Hazard Symbols |    GHS03;GHS06;GHS07 Danger Details GHS03;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H272-H301-H302-H319-H332 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P210-P220-P261-P264-P264+P265-P270-P271-P280-P301+P316-P301+P317-P304+P340-P305+P351+P338-P317-P321-P330-P337+P317-P370+P378-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 1446 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Barium nitrate is a chemical compound with the formula Ba(NO₃)₂, commonly used in pyrotechnics and as an oxidizing agent in various chemical reactions. It was first synthesized in the 19th century as part of the growing interest in barium compounds and their applications in industrial processes. Barium nitrate is an inorganic salt composed of barium cations (Ba²⁺) and nitrate anions (NO₃⁻), and it has found use in a variety of fields ranging from fireworks to the chemical industry. The discovery of barium nitrate can be traced to early research into the compounds of barium, a chemical element that was isolated in 1808 by Sir Humphry Davy. However, it was not until later that the nitrate form of barium was isolated and studied for its unique properties. By reacting barium carbonate or barium hydroxide with nitric acid, barium nitrate is formed, typically as a white, crystalline solid. The compound is highly soluble in water, making it useful in various aqueous solutions and industrial applications. One of the most prominent applications of barium nitrate is in the manufacture of pyrotechnic compositions, particularly in fireworks and flares. When used in fireworks, barium nitrate contributes to the green coloration observed during combustion, a result of the barium ions emitting light when heated. The compound’s role as an oxidizer in these formulations allows for the sustained combustion of other materials, contributing to the intensity and longevity of the pyrotechnic display. In addition to its use in fireworks, barium nitrate is also employed in the production of special glass, where it helps to improve the thermal properties and refractive index of the material. In the chemical industry, barium nitrate serves as an oxidizing agent in a variety of reactions. It is used in the production of other barium compounds, such as barium chromate, which is utilized in the manufacture of pigments and corrosion-resistant coatings. Additionally, it is involved in the preparation of chemicals for the automotive and electronics industries, where it can be used in the synthesis of materials that require controlled oxidation. Barium nitrate is also used in the production of certain types of ceramics, where it aids in achieving the desired chemical composition and properties in the final product. It has been studied for its potential to improve the performance of thermally stable ceramics used in high-temperature applications. In addition to its industrial uses, barium nitrate is sometimes used in laboratory settings as a reagent in analytical chemistry. Despite its many uses, barium nitrate must be handled with care, as it is toxic and can cause harmful effects if ingested or inhaled. It is classified as a hazardous substance and is subject to regulations regarding its handling, storage, and disposal. Proper safety measures are necessary when working with barium nitrate to avoid exposure to its potentially dangerous properties. In conclusion, barium nitrate is a versatile chemical compound with significant applications in pyrotechnics, chemical synthesis, glass manufacturing, and ceramics. Its ability to act as an oxidizing agent and contribute to color production in fireworks has made it a staple in the pyrotechnic industry, while its use in other industrial applications continues to highlight its importance in modern chemistry. References 2001. Rapid and sensitive determination of radiocesium (Cs-135, Cs-137) in the presence of excess barium by electrothermal vaporization-inductively coupled plasma-mass spectrometry (ETV-ICP-MS) with potassium thiocyanate as modifier. Fresenius' Journal of Analytical Chemistry, 370(5). DOI: 10.1007/s002160000678 2014. A review of the health impacts of barium from natural and anthropogenic exposure. Environmental Geochemistry and Health, 36(4). DOI: 10.1007/s10653-014-9622-7 2020. Characterisation of gunshot residues from non-toxic ammunition and their persistence on the shooter’s hands. International Journal of Legal Medicine, 134(5). DOI: 10.1007/s00414-020-02261-9 |
| Market Analysis Reports |