Lanthanum chloride heptahydrate was discovered after the isolation of lanthanum in the early 19th century. Swedish chemist Carl Gustav Mosander first discovered lanthanum in 1839 while studying cerium minerals. As research on rare earth elements progressed, lanthanum chloride and its hydrates, including the heptahydrate, were synthesized and studied for their unique properties and potential uses. Lanthanum chloride heptahydrate is a white crystalline solid that is highly soluble in water, forming a clear, colorless solution.
Lanthanum chloride heptahydrate is used as a catalyst in a variety of organic synthesis reactions. Its catalytic properties can be used to increase reaction rates and selectivity in processes such as polymerization, alkylation, and oxidation. Lanthanum chloride heptahydrate is used as a precursor to the synthesis of other lanthanum compounds, including oxides and mixed metal catalysts, which are used in the chemical and petrochemical industries to produce fuels, chemicals, and materials.
Lanthanum chloride heptahydrate is used in the production of specialized optical materials such as glass and ceramics that require high refractive index and transparency. Lanthanum-containing glasses are used in camera lenses, optical instruments, and optical fibers to improve image quality and light transmission. Lanthanum chloride heptahydrate is incorporated into materials used in electronics, such as phosphors and scintillators, which are essential for displays, lighting, and radiation detection. Lanthanum-based scintillators enhance the performance of X-ray and gamma-ray detectors used in medical imaging and security systems.
Lanthanum chloride heptahydrate is used to prepare high-temperature superconductors, particularly those containing rare earth elements. These superconductors can be used in advanced technologies, including magnetic resonance imaging (MRI), particle accelerators, and power transmission systems.
In biological research, lanthanum chloride heptahydrate is used as a tool to study calcium channels and signaling pathways. It acts as a calcium channel blocker, allowing researchers to study the role of calcium in cellular processes and disease. In the field of radiotherapy, lanthanum compounds are being studied for their potential to enhance the effectiveness of cancer radiotherapy. Lanthanum chloride's role in radiosensitization is being studied to improve treatment outcomes.
Lanthanum chloride heptahydrate is used in water treatment to precipitate phosphates, which reduces nutrient levels in water bodies and helps prevent algal blooms and eutrophication. Lanthanum trichloride heptahydrate is used to develop pollution control and environmental remediation materials, particularly to capture and neutralize pollutants in industrial emissions and waste streams.
References
1980. Extraction of hyaluronic acid from rabbit skin with lanthanum chloride. The Tohoku Journal of Experimental Medicine, 132(3).
DOI: 10.1620/tjem.132.337
2020. Use of lanthanum for water treatment A matter of concern? Chemosphere, 239.
DOI: 10.1016/j.chemosphere.2019.124780
2022. Biophysical interaction between lanthanum chloride and (CG)n or (GC)n repeats: A reversible B-to-Z DNA transition. International Journal of Biological Macromolecules, 216.
DOI: 10.1016/j.ijbiomac.2022.07.020
|


































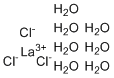


 GHS05;GHS07;GHS09 Danger Details
GHS05;GHS07;GHS09 Danger Details