Online Database of Chemicals from Around the World
| Shaanxi Kaida Chemcial Engineering Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kd-chem.com | |||
 | +86 (917) 331-7143 332-2918 | |||
 | +86 (917) 331-0616 | |||
 | kaida@kd-chem.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2014 | ||||
| W.C.Heraeus GmbH | Germany | |||
|---|---|---|---|---|
 | www.heraeus-chemicalproducts.com | |||
 | +49 (6181) 355-707 | |||
 | +49 (6181) 355-914 | |||
 | info@heraeus.com | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal chlorides and salts |
|---|---|
| Name | Iridium tetrachloride |
| Synonyms | tetrachloroiridium |
| Molecular Structure | 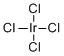 |
| Molecular Formula | IrCl4 |
| Molecular Weight | 334.03 |
| CAS Registry Number | 10025-97-5 |
| EC Number | 233-048-8 |
| SMILES | Cl[Ir](Cl)(Cl)Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Iridium tetrachloride, with the chemical formula IrCl4, is an inorganic compound consisting of iridium in the +4 oxidation state coordinated by four chloride ligands. It is a dark-colored solid, typically black or dark brown, and is highly hygroscopic and reactive. IrCl4 is primarily used as a precursor in the preparation of other iridium compounds and as a starting material in synthetic inorganic and organometallic chemistry. The study of iridium chlorides dates back to the 19th century when platinum group metals were systematically explored following their separation from native platinum ores. Iridium, discovered in 1803 by Smithson Tennant, was found to form various chlorinated species, with the tetrachloride recognized as a distinct oxidation state (+4) compound. However, due to the rarity and high cost of iridium, its chlorides were not widely produced or investigated until more recent advances in coordination chemistry and catalysis highlighted their significance. Iridium tetrachloride is most commonly prepared by the chlorination of iridium metal at elevated temperatures or by the decomposition of iridium hexachloride (H2IrCl6) under controlled thermal conditions. Another route involves treating iridium(III) chloride or iridium dioxide with chlorine gas or chlorinating agents in the presence of hydrochloric acid. The resulting IrCl4 is sensitive to moisture and decomposes slowly in air, releasing chlorine and forming lower oxidation state species such as IrCl3. As a coordination compound, IrCl4 can be used to synthesize a variety of iridium complexes. It reacts with ligands such as phosphines, nitrogen-donor molecules, or carbonyls to form stable organometallic and coordination compounds, many of which exhibit interesting electronic and catalytic properties. These reactions often involve reduction of Ir(IV) to Ir(III) or Ir(I), depending on the ligand and reaction conditions. In catalysis, iridium compounds derived from IrCl4 are used in a number of homogeneous and heterogeneous catalytic processes. Although IrCl4 itself is not a common catalyst, it serves as a source of iridium in the preparation of active catalysts used in hydrogenation, hydrosilylation, water oxidation, and C–H bond activation. Iridium-based catalysts are valued for their high activity and stability, especially under oxidative conditions. Iridium tetrachloride also finds application in materials science. It has been used in the preparation of iridium thin films and nanoparticles, often by thermal decomposition or reduction. These materials are studied for their use in electronic devices, sensors, and as components of high-temperature and corrosion-resistant coatings. Analytical techniques such as X-ray diffraction, infrared spectroscopy, and thermogravimetric analysis are used to characterize IrCl4. In its solid state, IrCl4 typically forms polymeric structures, with each iridium atom octahedrally coordinated by chloride ligands, some of which bridge to neighboring iridium atoms. These structural features influence its reactivity and solubility. The compound is soluble in concentrated hydrochloric acid and some organic solvents, forming chlorometalate complexes in solution. Due to the toxicity and environmental concerns associated with heavy metal halides, appropriate precautions are necessary when handling iridium tetrachloride. It should be used in a well-ventilated fume hood with gloves and protective clothing. Waste containing iridium compounds must be treated according to local regulations for hazardous materials. In summary, iridium tetrachloride is a key compound in iridium coordination chemistry and is used as a precursor in the synthesis of diverse iridium complexes. Its applications extend to catalysis and materials science, supported by its reactivity and the unique properties of the iridium center in oxidation state +4. References 1986. 35Cl NQR spectra of osmium and iridium chlorochalcogen complexes. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 35(6). DOI: 10.1007/bf00956616 2002. Extraction of Iridium(IV) from Hydrochloric Acid Solution with Bis(piperidinoethylthioethyl)-1-phenyl-1-ethanone. Russian Journal of Applied Chemistry, 75(6). DOI: 10.1023/a:1020368209117 2022. Synthesis of Ir-CaO Complex Oxides and Their Solubilities in Hydrochloric Acid. Journal of Sustainable Metallurgy, 8(4). DOI: 10.1007/s40831-022-00643-4 |
| Market Analysis Reports |