Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shanghai Boka-chem Tech Inc. | China | |||
|---|---|---|---|---|
 | www.bokachem.com | |||
 | +86 (21) 6478-0600 | |||
 | +86 (21) 6459-2060 | |||
 | lidong@bokachem.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neijiang Noble Materil Technology. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njsnoble.com | |||
 | +86 (832) 220-6225 | |||
 | +86 (832) 220-6225 | |||
 | 3080634228@qq.com | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Kangchi Chemical Industry (shenzhen) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.konchie.com | |||
 | +86 (755) 8257-4253 | |||
 | +86 (755) 3315-1032 | |||
 | kangchi@konchie.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Jinan Hanyu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hanyuchem.com | |||
 | +86 (531) 6995-4981 | |||
 | sales@hanyuchem.com | |||
 | QQ Chat | |||
 | WeChat: dwyane-wang | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Pressure Chemical Company | USA | |||
|---|---|---|---|---|
 | www.presschem.com | |||
 | +1 (800) 722-5247 (412) 682-5882 | |||
 | +1 (412) 682-5864 | |||
 | contact_us@presschem.com | |||
| Chemical manufacturer since 1964 | ||||
| Colonial Metals, Inc. | USA | |||
|---|---|---|---|---|
 | www.colonialmetals.com | |||
 | +1 (410) 398-7200 (800) 962-1537 | |||
 | +1 (410) 398-2918 | |||
 | sales@colonialmetals.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Metalor Technologies Group | China | |||
|---|---|---|---|---|
 | www.metalor.com | |||
 | +86 (512) 6593-6181 +86 (21) 5442-3060 | |||
 | +86 (512) 6593-6171 / +86 (21) 5442-0790 | |||
 | hr.cn@metalor.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| W.C.Heraeus GmbH | Germany | |||
|---|---|---|---|---|
 | www.heraeus-chemicalproducts.com | |||
 | +49 (6181) 355-707 | |||
 | +49 (6181) 355-914 | |||
 | info@heraeus.com | |||
| Chemical manufacturer | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| Classification | Food additive >> Nutrition supplements >> Inorganic salts |
|---|---|
| Name | Dipotassium tetrachloroplatinate |
| Synonyms | Potassium tetrachloroplatinate(II) |
| Molecular Structure | 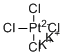 |
| Molecular Formula | K2PtCl4 |
| Molecular Weight | 415.08 |
| CAS Registry Number | 10025-99-7 |
| EC Number | 233-050-9 |
| SMILES | Cl[Pt-2](Cl)(Cl)Cl.[K+].[K+] |
| Density | 3.38 g/mL (Expl.) |
|---|---|
| Melting point | (Decomposes) (Expl.) |
| Solubility | 10 g/L (20 °C) (Expl.) |
| Hazard Symbols |      GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H290-H301-H315-H317-H318-H334-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P233-P234-P260-P261-P264-P264+P265-P270-P271-P272-P273-P280-P284-P301+P316-P302+P352-P304+P340-P305+P354+P338-P317-P321-P330-P332+P317-P333+P317-P342+P316-P362+P364-P390-P391-P403-P405-P406-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3288 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Dipotassium tetrachloroplatinate, with the chemical formula K2PtCl4, is a well-known platinum(II) coordination compound. Its discovery can be traced back to the 19th century during the systematic investigation of platinum salts. Early studies of platinum chemistry were driven by the metal’s remarkable resistance to corrosion and its unique catalytic properties. Dipotassium tetrachloroplatinate was synthesized by dissolving elemental platinum in aqua regia to form chloroplatinic acid (H2PtCl6), followed by neutralization with potassium chloride, leading to the formation of K2PtCl4 upon crystallization. The crystallographic characterization of dipotassium tetrachloroplatinate revealed that it forms red to orange crystalline solids that are soluble in water but less soluble in alcohols. The platinum center is coordinated in a square planar geometry by four chloride ligands, a typical arrangement for platinum(II) complexes. This structural feature was confirmed by X-ray crystallography and spectroscopic analyses, providing a deeper understanding of the coordination chemistry of platinum. Dipotassium tetrachloroplatinate has significant applications, particularly in the synthesis of other platinum compounds. It serves as an important starting material for the preparation of a wide range of platinum complexes, including both platinum(II) and platinum(IV) species. Many organoplatinum compounds used in catalysis, materials science, and medicinal chemistry are synthesized through substitution reactions involving K2PtCl4. One of the most notable applications of dipotassium tetrachloroplatinate is in the production of cisplatin, cis-diamminedichloroplatinum(II), a groundbreaking anticancer drug. The preparation of cisplatin involves the reaction of K2PtCl4 with ammonia under controlled conditions, selectively replacing two chloride ligands with ammine ligands in a cis configuration. Cisplatin’s discovery and clinical success highlighted the importance of K2PtCl4 as a crucial precursor in pharmaceutical chemistry. In addition to medicinal chemistry, dipotassium tetrachloroplatinate is employed in electroplating processes, where platinum coatings are valued for their durability, electrical conductivity, and resistance to tarnishing. It is also used as a reagent in analytical chemistry, particularly in gravimetric analysis, to precipitate potassium as potassium hexachloroplatinate(VI) for quantitative determination. Catalysis represents another significant area where dipotassium tetrachloroplatinate finds utility. It is used to prepare various platinum catalysts that are active in hydrogenation, hydrosilylation, and oxidation reactions. The ability to tailor the ligand environment around platinum through substitution of the chloride ligands in K2PtCl4 allows for the fine-tuning of catalyst properties to suit specific reactions. Furthermore, dipotassium tetrachloroplatinate has applications in materials science. It is used to produce platinum nanoparticles and thin films, which are integral components in the development of sensors, fuel cells, and other electronic devices. The compound’s aqueous solubility and chemical reactivity make it suitable for solution-based methods of nanoparticle synthesis, such as chemical reduction processes. Overall, dipotassium tetrachloroplatinate remains a substance of great importance in various fields of chemistry and industry, owing to its well-characterized structure, reliable reactivity, and versatility as a precursor to a multitude of platinum-based materials and compounds. References 1997. Immunostimulatory Effects of Platinum Compounds: Correlation between Sensitizing Properties in vivo and Modulation of Receptor-Mediated Endocytosis in vitro. International Archives of Allergy and Immunology, 112(2). DOI: 10.1159/000237442 2008. Speciation of inorganic platinum-chloride complexes in spiked environmental samples by SPE and ICP-AES. Analytica Chimica Acta, 616(1). DOI: 10.1016/j.aca.2008.03.037 2013. Occupational Immediate-Type Allergic Asthma due to Potassium Tetrachloroplatinate in Production of Cytotoxic Drugs. Advances in Experimental Medicine and Biology, 755. DOI: 10.1007/978-94-007-4546-9_6 |
| Market Analysis Reports |