Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Struchem Co., Ltd | China | |||
|---|---|---|---|---|
 | www.struchem.com | |||
 | +86 (21) 3353-8390 | |||
 | +86 (21) 3208-2820 | |||
 | sales@struchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Aromsyn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aromsyn.com | |||
 | +86 (571) 8558-5865 +86 13336024896 | |||
 | +86 (571) 8558-5865 | |||
 | service@aromsyn.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Nanjing Fred Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fredbio.com | |||
 | +86 (25) 8469-6168 | |||
 | Austin@fredchem.cn | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: NJFred01 | |||
 | WhatsApp:+86 17302533743 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | 2'-Bromo-5'-fluoroacetophenone |
| Synonyms | 1-(2-bromo-5-fluorophenyl)ethanone |
| Molecular Structure | 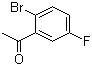 |
| Molecular Formula | C8H6BrFO |
| Molecular Weight | 217.04 |
| CAS Registry Number | 1006-33-3 |
| EC Number | 600-114-6 |
| SMILES | CC(=O)C1=C(C=CC(=C1)F)Br |
| Density | 1.5±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 251.1±25.0 °C 760 mmHg (Calc.)* |
| Flash point | 105.7±23.2 °C (Calc.)* |
| Index of refraction | 1.534 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
2'-Bromo-5'-fluoroacetophenone is an aromatic ketone that contains both bromine and fluorine substituents on the phenyl ring of an acetophenone framework. It is classified as a halogenated acetophenone derivative, with the halogens positioned meta and para to the ketone group. The molecular formula is C8H6BrFO, and its structure includes a bromo substituent at the 2' position and a fluoro substituent at the 5' position on the phenyl ring, relative to the acetyl group. This compound is primarily used as an intermediate in organic synthesis, particularly in the preparation of pharmaceutical and agrochemical compounds. The presence of both electron-withdrawing halogens and a reactive carbonyl group makes it suitable for further functionalization through substitution or coupling reactions, such as Suzuki or Heck cross-couplings. These transformations allow for the introduction of a wide variety of functional groups, enabling the synthesis of structurally diverse molecules. The synthesis of 2'-bromo-5'-fluoroacetophenone can be achieved by directed halogenation of appropriately substituted acetophenones or by selective halogen exchange reactions using starting materials such as 2'-bromoacetophenone followed by electrophilic fluorination. The regioselectivity is typically governed by electronic and steric factors, which are critical in ensuring the correct substitution pattern on the aromatic ring. In applied research, derivatives of halogenated acetophenones, including compounds like 2'-bromo-5'-fluoroacetophenone, are evaluated for their biological activity. While 2'-bromo-5'-fluoroacetophenone itself is not widely documented as a final active pharmaceutical ingredient, it serves as a valuable building block in the preparation of compounds that may exhibit antimicrobial, anti-inflammatory, or enzyme-inhibitory properties, depending on the final structure. Its reactivity profile and structural features make it a convenient candidate in medicinal chemistry and materials science where halogenated aromatic ketones are frequently employed. The use of such intermediates is well-established in literature, and their importance lies in enabling the synthesis of compounds with high selectivity and desirable physicochemical characteristics. References none |
| Market Analysis Reports |