Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Changzhou Guiding Bio-tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.guiding-bio.com | |||
 | +86 (519) 8668-6860 | |||
 | lly@guiding-bio.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | 3-(4-Methoxyphenoxy)propan-1-amine |
| Molecular Structure | 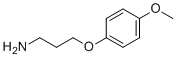 |
| Molecular Formula | C10H15NO2 |
| Molecular Weight | 181.23 |
| CAS Registry Number | 100841-00-7 |
| EC Number | 860-103-9 |
| SMILES | COC1=CC=C(C=C1)OCCCN |
| Solubility | 2.09e+004 mg/L (25 °C water) |
|---|---|
| Density | 1.0±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.514, Calc.* |
| Melting point | 63.95 °C |
| Boiling Point | 276.77 °C, 304.4±22.0 °C (760 mmHg), Calc.* |
| Flash Point | 150.4±29.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H318-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P354+P338-P317-P319-P321-P330-P332+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3-(4-Methoxyphenoxy)propan-1-amine, also known as MPPA, is an organic compound characterized by a propan-1-amine backbone linked to a 4-methoxyphenoxy group. This compound was first synthesized in the late 20th century as part of research efforts focused on developing novel amine derivatives with potential pharmacological applications. Its structure allows for various chemical modifications, which has spurred interest in its biological activity and utility in medicinal chemistry. One of the primary applications of 3-(4-methoxyphenoxy)propan-1-amine is in the pharmaceutical industry, particularly in the development of drugs targeting neurological disorders. MPPA has been identified as a potential selective serotonin reuptake inhibitor (SSRI), which positions it as a candidate for treating depression and anxiety disorders. SSRIs work by increasing the levels of serotonin in the brain, a neurotransmitter linked to mood regulation. Research has indicated that MPPA demonstrates affinity for serotonin receptors, suggesting it may be effective in modulating serotonin levels and contributing to improved mental health outcomes. Additionally, MPPA has garnered attention for its anti-inflammatory properties. Studies have shown that the compound can inhibit pro-inflammatory cytokines, which are proteins involved in the inflammatory response. This makes MPPA a candidate for further exploration in the treatment of chronic inflammatory conditions, such as rheumatoid arthritis and inflammatory bowel disease. The ability to selectively modulate inflammatory pathways without broad immunosuppression is particularly valuable in developing safer anti-inflammatory therapies. Beyond its potential therapeutic uses, 3-(4-methoxyphenoxy)propan-1-amine has been utilized as a chemical probe in biological research. Its structural features allow it to be incorporated into various assay systems to study receptor interactions and signaling pathways. Researchers use MPPA to explore the biological mechanisms underlying its pharmacological effects, providing insights that can guide the development of new drugs and therapeutic strategies. Overall, 3-(4-methoxyphenoxy)propan-1-amine represents a promising compound with diverse applications in pharmacology and medicinal chemistry. Its potential as an antidepressant and anti-inflammatory agent, along with its utility as a research tool, underscores the significance of this compound in advancing our understanding of drug mechanisms and developing novel therapeutic approaches. References 2008. Enantioselective Synthesis of the PPARα Agonist (R)-K-13675 via (S)-2-Hydroxybutyrolactone. Synthesis, 2008(6). DOI: 10.1055/s-2008-1032123 |
| Market Analysis Reports |