Lead(II) iodide (PbI2) is an inorganic compound consisting of lead in the +2 oxidation state and iodide ions. It is well known for its striking golden-yellow crystalline appearance, which has earned it the common name “golden yellow lead iodide.” PbI2 crystallizes in a layered structure, where sheets of lead ions are sandwiched between layers of iodide ions. This layered arrangement gives rise to its distinctive optical and electronic properties.
The compound was first described in the early 19th century during systematic investigations into halogen salts of heavy metals. Lead iodide was commonly prepared by adding a soluble iodide salt, such as potassium iodide, to an aqueous solution of a soluble lead salt, such as lead nitrate or lead acetate. The reaction produces a bright yellow precipitate of PbI2, which quickly became a standard demonstration in chemistry education to illustrate precipitation reactions and solubility equilibria.
In terms of applications, lead(II) iodide has found use in several areas of materials science and technology. One of its most significant modern roles is as a precursor for the synthesis of lead halide perovskites, which are widely investigated for use in next-generation solar cells, light-emitting diodes, lasers, and photodetectors. In these applications, PbI2 is reacted with organic halides such as methylammonium iodide or formamidinium iodide to form perovskite thin films with high optical absorption and excellent charge transport properties. These perovskite materials have enabled solar cells with efficiencies rivaling or surpassing traditional silicon-based devices.
PbI2 also has a history of application in radiation detection. Crystals of lead iodide exhibit semiconducting and scintillating properties, allowing them to detect gamma rays and X-rays. Because PbI2 is composed of heavy atoms, it has high stopping power for high-energy photons, making it suitable for compact radiation detectors and medical imaging devices. Research into single-crystal growth techniques has advanced its potential use in portable and room-temperature radiation detectors.
In traditional chemistry education, the precipitation of lead iodide continues to serve as a classroom experiment for illustrating recrystallization and crystallography. When recrystallized slowly from hot water, PbI2 forms striking golden hexagonal plate crystals, which are visually appealing and provide a tangible example of crystal growth processes.
Despite its scientific and technological importance, PbI2 is hazardous due to its lead content. Lead compounds are toxic and can cause serious health effects, particularly on the nervous system, kidneys, and developmental processes. Consequently, handling of PbI2 requires careful attention to laboratory safety protocols, and its use in consumer applications is limited by strict regulations.
In summary, lead(II) iodide is a historically well-known compound with characteristic golden-yellow crystals and a layered crystal structure. From its early role in classical inorganic chemistry to its central position in modern materials research, PbI2 exemplifies the scientific importance of simple inorganic compounds. Today, its primary applications lie in the preparation of perovskite semiconductors and in the development of radiation detection materials, though its toxicity continues to necessitate strict safety measures.
|


























































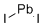


 GHS07;GHS08;GHS09 Danger Details
GHS07;GHS08;GHS09 Danger Details