Online Database of Chemicals from Around the World
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic iron |
|---|---|
| Name | Ferrocene |
| Synonyms | Di-2,4-cyclopentadien-1-yliron; Biscyclopentadienyliron; Dicyclopentadienyliron |
| Molecular Structure | 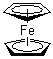 |
| Molecular Formula | C10H10Fe |
| Molecular Weight | 186.03 |
| CAS Registry Number | 102-54-5 |
| EC Number | 203-039-3 |
| SMILES | [CH-]1C=CC=C1.[CH-]1C=CC=C1.[Fe+2] |
| Melting point | 173-176 °C |
|---|---|
| Sublimation | 100 °C |
| Boiling point | 249 °C |
| Water solubility | practically insoluble |
| Hazard Symbols |     GHS02;GHS07;GHS08;GHS09 Danger Details GHS02;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H228H302+H332-H302-H332-H360-H373-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P210-P240-P241-P260-P261-P264-P270-P271-P273-P280-P301+P317-P304+P340-P317-P318-P319-P330-P370+P378-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1325 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Ferrocene, an organometallic compound with the formula Fe(C₅H₅)₂, stands as one of the most iconic molecules in the field of chemistry due to its unique structure and versatile applications. Discovered in 1951, ferrocene was the first metallocene compound to be synthesized, marking the advent of a new class of organometallic compounds known as sandwich compounds. This discovery significantly advanced the understanding of bonding between transition metals and organic ligands, leading to a range of applications in materials science, catalysis, and medicine. The synthesis of ferrocene occurred almost accidentally when researchers were investigating the reaction between cyclopentadienyl magnesium bromide and ferric chloride. The resulting compound exhibited unexpected stability and a distinctive orange color. Upon structural analysis, it was revealed that ferrocene consists of two cyclopentadienyl anions (C₅H₅⁻) bonded symmetrically to a central iron (Fe²⁺) atom. This structure, where the iron atom is sandwiched between two parallel cyclopentadienyl rings, challenged the existing theories of bonding and prompted the development of the concept of aromaticity in organometallic chemistry. The unique structure of ferrocene has endowed it with several remarkable properties, including thermal stability, chemical resistance, and ease of substitution at the cyclopentadienyl rings. These characteristics have led to its widespread use in various applications. In materials science, ferrocene is a key building block for the synthesis of conducting polymers and molecular electronic devices. Its ability to undergo reversible redox reactions makes it an ideal candidate for use in batteries, sensors, and other electrochemical applications. In the field of catalysis, ferrocene derivatives have been extensively studied and utilized. The iron center in ferrocene can be functionalized to create catalysts that are highly effective in a range of organic transformations, including hydrogenation, polymerization, and cross-coupling reactions. These ferrocene-based catalysts offer advantages such as high activity, selectivity, and the ability to operate under mild conditions, making them valuable in both academic research and industrial processes. Ferrocene and its derivatives have also found applications in medicine. The compound's stability and low toxicity make it suitable for use in drug design and development. Ferrocene-containing drugs have been explored for their potential as anticancer agents, leveraging the redox properties of the iron center to generate reactive oxygen species that can selectively target cancer cells. Additionally, ferrocene derivatives are being investigated for their use in diagnostic imaging and as therapeutic agents in various diseases. The discovery of ferrocene not only expanded the scope of organometallic chemistry but also paved the way for the development of numerous other metallocenes, each with its own unique properties and applications. The versatility of ferrocene continues to inspire research across multiple disciplines, from materials science to medicine, underscoring its enduring significance in modern chemistry. References 2024. Ansa-Ferrocene Derivatives as Potential Therapeutics. Molecules, 29(20). DOI: 10.3390/molecules29204903 2024. Repurposing of monocyclic β-lactams as anti-inflammatory agents - The case of new ferrocene-azetidin-2-one hybrids. European Journal of Medicinal Chemistry, 280. DOI: 10.1016/j.ejmech.2024.116910 2024. A Novel Ferrocene-Linked Thionine as a Dual Redox Mediator for the Electrochemical Detection of Dopamine and Hydrogen Peroxide. Biosensors, 14(9). DOI: 10.3390/bios14090448 |
| Market Analysis Reports |