Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Ningbo Sinotop Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinotopchem.com | |||
 | +86 (574) 8823-7971 | |||
 | +86 (574) 8823-7323 | |||
 | mia@sinotopchem.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | 2-Oxohexamethylenimine |
| Synonyms | epsilon-Caprolactam; Aza-2-cycloheptanone |
| Molecular Structure | 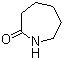 |
| Molecular Formula | C6H11NO |
| Molecular Weight | 113.16 |
| CAS Registry Number | 105-60-2 |
| EC Number | 203-313-2 |
| SMILES | C1CCC(=O)NCC1 |
| Density | 1.0±0.1 g/cm3, Calc.*, 1.01 g/mL (Expl.) |
|---|---|
| Melting point | 68-71 °C (Expl.) |
| Index of Refraction | 1.446, Calc.* |
| Boiling Point | 272.5 °C (760 mmHg), Calc.*, 300.8-303.5 °C (Expl.) |
| Flash Point | 136.7±3.4 °C, Calc.*, 152 °C (Expl.) |
| Water solubility | 53.0 g/100g (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H332-H302-H335-H315-H319 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
2-Oxohexamethylenimine, also known as ε-caprolactam, is an organic compound with the molecular formula C&sub6;H&sub11;NO. It is a white, crystalline solid that is hygroscopic and has a slight odor. This cyclic amide, or lactam, is primarily recognized for its role as the monomer in the production of Nylon 6, a synthetic polymer that is widely used in various industrial applications. The discovery and industrial production of ε-caprolactam can be traced back to the early 20th century. The development of its manufacturing processes has evolved significantly over the years. One of the most common methods for producing ε-caprolactam is the Beckmann rearrangement of cyclohexanone oxime. In this process, cyclohexanone is first converted to cyclohexanone oxime using hydroxylamine, and the oxime undergoes acid-catalyzed rearrangement to yield ε-caprolactam. This method has been widely adopted due to its efficiency and high yield. Other production methods include the reaction of cyclohexane with nitric acid or oxidation processes involving benzene derivatives. ε-Caprolactam is predominantly used in the production of Nylon 6, a polyamide that is valued for its mechanical strength, elasticity, resistance to wear, and chemical stability. Nylon 6 is extensively employed in the textile industry for manufacturing fibers used in fabrics, carpets, and industrial yarns. Due to its durability and resistance to environmental factors, it is also used in the production of engineering plastics, which are found in automotive parts, electrical components, and various consumer goods. In addition to its use in polymer production, ε-caprolactam is utilized in the manufacture of synthetic leather, adhesives, coatings, and films. The compound's ability to form strong, durable materials makes it essential in applications that require high-performance synthetic materials. Furthermore, ε-caprolactam-based products are used in food packaging and medical applications due to their non-toxicity and compliance with safety regulations. From a chemical perspective, ε-caprolactam is a polar compound that exhibits good solubility in water and many organic solvents. It has a melting point in the range of 68-71°C and a boiling point of approximately 136-138°C under reduced pressure. The compound's hygroscopic nature means that it readily absorbs moisture from the air, which can influence its handling and storage conditions. To ensure its stability, it is typically stored in airtight containers under dry conditions. Handling and safety considerations are important when working with ε-caprolactam. While it is not highly toxic, it can cause irritation to the skin, eyes, and respiratory tract upon direct exposure. Appropriate protective measures, such as gloves, goggles, and adequate ventilation, are recommended when handling this compound in industrial and laboratory settings. The production of ε-caprolactam is an integral part of the global chemical industry, with large-scale manufacturing facilities operating in various countries. Environmental considerations have led to the development of more sustainable production methods, including catalytic oxidation and biotechnological approaches, to reduce waste and improve efficiency. Advances in recycling technologies have also contributed to the circular economy of polyamide materials, allowing for the recovery and reuse of caprolactam-derived polymers. ε-Caprolactam remains one of the most important industrial chemicals, with its primary application in Nylon 6 synthesis driving its global demand. Its versatility, chemical stability, and mechanical properties make it a key material in modern manufacturing, ensuring its continued relevance across multiple industries. References 2024. Alkaline hydrothermal cracking effect and substance transformation characteristics of caprolactam-containing sludge. Environmental pollution (Barking, Essex : 1987), 364. DOI: 10.1016/j.envpol.2024.124955 2024. Study of polymer component migrated in medicinal product from transportation packaging component: A systematic assessment beyond regulatory expectations. Journal of Pharmaceutical Sciences, 113(12). DOI: 10.1016/j.xphs.2024.09.021 2024. Ecological risk assessment and characterization of microplastics in the beach sediments of southeast coast of India. Environmental Monitoring and Assessment, 196(12). DOI: 10.1007/s10661-024-13379-3 |
| Market Analysis Reports |