Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| VanDeMark Chemical Inc. | USA | |||
|---|---|---|---|---|
 | www.vandemark.com | |||
 | +1 (716) 433-6764 | |||
 | +1 (716) 433-2850 | |||
 | sales@vandemark.com | |||
| Chemical manufacturer since 1951 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Acid ester compound |
|---|---|
| Name | Oxydiethylene bis(chloroformate) |
| Synonyms | Diethylene glycol bis-chloroformate |
| Molecular Structure | 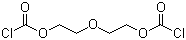 |
| Molecular Formula | C6H8Cl2O5 |
| Molecular Weight | 231.03 |
| CAS Registry Number | 106-75-2 |
| EC Number | 203-430-9 |
| SMILES | C(COC(=O)Cl)OCCOC(=O)Cl |
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H318-H411 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P272-P273-P280-P301+P317-P302+P352-P305+P354+P338-P317-P321-P330-P332+P317-P333+P317-P362+P364-P391-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Oxydiethylene bis(chloroformate) is a chemical compound commonly used as a reagent in organic synthesis, particularly in the production of polyurethanes and other polymers. Its structure consists of two chloroformate groups connected by an ethylene glycol linker, giving it the molecular formula C5H8Cl2O5. The compound is a colorless liquid and is reactive toward nucleophiles, making it useful for introducing chloroformate functionalities into other molecules. The discovery of oxydiethylene bis(chloroformate) was part of the development of reagents that could efficiently form carbonates and carbamates. Chloroformates, in general, are versatile intermediates that can react with alcohols and amines to form esters and carbamates, respectively. Oxydiethylene bis(chloroformate) has the advantage of having two reactive chloroformate groups, allowing it to link two molecules or to participate in polymerization reactions, forming linear or crosslinked polymers. One of the main applications of oxydiethylene bis(chloroformate) is in the synthesis of polyurethanes. In this process, the compound reacts with diamines or diols to form polymer chains through the formation of urethane or carbonate linkages. The resulting polyurethanes are used in a variety of applications, including in the production of foams, coatings, and adhesives. The bifunctional nature of oxydiethylene bis(chloroformate) makes it particularly useful in creating polymers with controlled architectures, contributing to the development of materials with specific mechanical and chemical properties. In addition to its role in polymer chemistry, oxydiethylene bis(chloroformate) is also used as a reagent in the modification of proteins and peptides. Its ability to introduce chloroformate groups allows for the functionalization of biomolecules, enabling the attachment of various labels, crosslinkers, or other functional groups. This has made it a valuable tool in bioconjugation and protein engineering, where precise chemical modifications are often required. Oxydiethylene bis(chloroformate) is also employed in the pharmaceutical industry for the synthesis of drug molecules. Its reactivity with amines allows it to be used in the preparation of carbamate-based drugs, which are known for their stability and biological activity. The compound's versatility in forming stable chemical bonds has made it a critical reagent in the development of various therapeutic agents. References 2013. Synthesis, characterization and properties of 3,3′-diethyl-1,1′-oxydiethylenedicarbonyl bis(thiourea). Research on Chemical Intermediates, 40(3), 1297-1307. DOI: 10.1007/s11164-013-1100-9 1995. Diethylene glycol bis(chloroformate). Toxicological Evaluations, 91-104. DOI: 10.1007/978-3-642-79169-7_5 1967. Zur Bestimmung von Allylchloroformiat und Diäthylenglykol-bis-chloroformiat in Diäthylenglykol-bis-allylcarbonat. Fresenius' Zeitschrift für analytische Chemie, 227(3), 161-166. DOI: 10.1007/bf00502279 |
| Market Analysis Reports |