Online Database of Chemicals from Around the World
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate |
|---|---|
| Name | 3-(4-Hydroxy-1-oxoisoindolin-2-yl)piperidine-2,6-dione |
| Synonyms | 3-(7-hydroxy-3-oxo-1H-isoindol-2-yl)piperidine-2,6-dione |
| Molecular Structure | 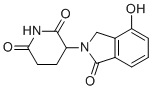 |
| Molecular Formula | C13H12N2O4 |
| Molecular Weight | 260.25 |
| CAS Registry Number | 1061604-41-8 |
| EC Number | 896-921-8 |
| SMILES | C1CC(=O)NC(=O)C1N2CC3=C(C2=O)C=CC=C3O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3-(4-Hydroxy-1-oxoisoindolin-2-yl)piperidine-2,6-dione, often abbreviated as hydroxythalidomide, is a derivative of thalidomide. This compound has potential in drug development, combining the structural elements of piperidine-2,6-dione and hydroxy-substituted isoindolinone, making it a promising molecule in medicinal chemistry. The discovery of hydroxythalidomide stems from the modification of thalidomide, a once controversial drug with teratogenic effects but significant therapeutic effects. Researchers aimed to create analogs with improved safety and enhanced biological activity. Further exploration revealed that hydroxythalidomide has a piperidine-2,6-dione core connected to a hydroxy-substituted isoindolinone, a structure that retains the pharmacological benefits of thalidomide while reducing side effects. Hydroxythalidomide is synthesized through organic reactions modified from thalidomide or its intermediates. The hydroxyl group at the 4-position of the isoindolinone ring introduces a key functional group that influences the compound's reactivity and interaction with biological targets. This modification enhances its solubility and increases its binding affinity to certain proteins, expanding its range of applications. Studies of the anti-inflammatory and immunomodulatory properties of hydroxythalidomide have found that, like thalidomide, it inhibits the production of tumor necrosis factor-α (TNF-α), a cytokine associated with inflammation. This makes it valuable in treating diseases such as rheumatoid arthritis and Crohn's disease by modulating immune responses and reducing inflammation. The compound has also been intensively studied for its anticancer potential. Hydroxythalidomide and its derivatives have shown efficacy in inhibiting the growth of certain cancer cells. They interfere with angiogenesis (the formation of new blood vessels), which is essential for tumor growth and metastasis. By blocking the blood supply to tumors, these compounds can limit cancer progression, making them promising drug candidates in oncology. In hematological diseases, hydroxythalidomide has shown therapeutic efficacy, particularly in multiple myeloma. Its ability to modulate the immune system and inhibit angiogenesis makes it an effective treatment for this cancer, in which abnormal plasma cells proliferate in the bone marrow. Emerging research suggests that hydroxythalidomide may have neuroprotective effects. Research is exploring its potential to treat neurodegenerative diseases such as Alzheimer's and Parkinson's by protecting neurons from inflammatory damage and oxidative stress. Hydroxythalidomide is a model for developing safer thalidomide analogs. By understanding its interactions, scientists can design new compounds with low teratogenicity while retaining or enhancing therapeutic effects. This approach aims to overcome thalidomide's historical shortcomings and safely harness its therapeutic potential. References 2023. Structural rationalization of GSPT1 and IKZF1 degradation by thalidomide molecular glue derivatives. RSC Medicinal Chemistry. DOI: 10.1039/d2md00347c 2013. Isosteric analogs of lenalidomide and pomalidomide: Synthesis and biological activity. Bioorganic & Medicinal Chemistry Letters. DOI: 10.1016/j.bmcl.2012.10.071 |
| Market Analysis Reports |