Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridazine |
|---|---|
| Name | 3,6-Dichloro-4-isopropylpyridazine |
| Molecular Structure | 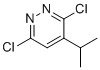 |
| Molecular Formula | C7H8Cl2N2 |
| Molecular Weight | 191.06 |
| CAS Registry Number | 107228-51-3 |
| SMILES | CC(C)C1=CC(=NN=C1Cl)Cl |
| Density | 1.274±0.06 g/cm3 |
|---|---|
| Boiling point | 278.7±35.0 °C |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details |
|---|---|
| Risk Statements | H301-H311-H331 Details |
| Safety Statements | P261-P264-P270-P271-P280-P302+P352-P304+P340-P310-P330-P361-P403+P233-P405-P501 Details |
| Transport Information | UN 2810 |
| SDS | Available |
|
3,6-Dichloro-4-isopropylpyridazine is a chlorinated pyridazine derivative, a class of heterocyclic compounds frequently studied for their diverse chemical reactivity and potential biological activity. Pyridazines are six-membered aromatic rings with two nitrogen atoms at the 1- and 2-positions, giving them unique properties that make them useful in various applications, particularly in the fields of agrochemicals and pharmaceuticals. The substitution pattern in 3,6-dichloro-4-isopropylpyridazine, with chlorine atoms at the 3- and 6-positions and an isopropyl group at the 4-position, enhances the molecule’s hydrophobicity and may contribute to its reactivity and stability. The discovery of chlorinated pyridazines such as 3,6-dichloro-4-isopropylpyridazine is tied to early research in heterocyclic chemistry, where scientists aimed to create modified ring structures that could exhibit desirable chemical and biological properties. Pyridazine derivatives, in particular, have been a focal point for developing herbicides, fungicides, and insecticides, owing to their potential to disrupt biochemical pathways in targeted pests without causing harm to the environment or non-target organisms. The addition of chlorine atoms is known to increase a compound’s persistence and bioactivity, which has made chlorinated pyridazines a key area of research in agriculture. The primary application of 3,6-dichloro-4-isopropylpyridazine is in agrochemical research and development, where it is used as a lead compound or intermediate in the synthesis of crop protection agents. Its structure allows for further modifications, such as additional halogenation or functionalization at various ring positions, which can lead to enhanced herbicidal or fungicidal properties. This compound has shown promise in preclinical evaluations as a building block for herbicides that target specific enzymes in weeds or as a fungicide for protecting crops from fungal pathogens. Researchers have also investigated derivatives of 3,6-dichloro-4-isopropylpyridazine as potential insecticides, focusing on their ability to interact with insect nervous systems selectively. In addition to agrochemical uses, pyridazine derivatives like 3,6-dichloro-4-isopropylpyridazine are studied for potential pharmaceutical applications. Pyridazine structures have shown activity in a variety of biological assays, including anti-inflammatory, anticancer, and antiviral testing. While 3,6-dichloro-4-isopropylpyridazine itself may not be used directly as a drug, its core structure serves as a valuable scaffold for designing analogs with therapeutic activity. Medicinal chemists often explore such compounds for developing enzyme inhibitors or ligands for biological receptors due to the electron-rich nitrogen atoms that enable binding interactions within biological systems. Furthermore, 3,6-dichloro-4-isopropylpyridazine is utilized in material science, especially in the development of specialized polymers and dyes. The pyridazine ring’s stability and chlorinated structure make it an attractive choice for designing materials that require resilience under extreme conditions. Some studies have focused on its incorporation into polymers for creating water-resistant or high-durability coatings, while others have explored its use in dye synthesis due to its ability to produce vibrant colors and resistance to fading. Overall, 3,6-dichloro-4-isopropylpyridazine exemplifies the versatility of pyridazine derivatives in multiple fields. From its role in crop protection to its potential applications in pharmaceuticals and material science, this compound continues to be a subject of interest in various research sectors. Its ease of modification and stability make it a valuable scaffold for further exploration in the development of new chemicals that could have significant impacts on agriculture, medicine, and industry. References 2019. Decarboxylative Alkylation. Synfacts. DOI: 10.1055/s-0039-1689844 2004. Introduction of Alkyl and Other Groups. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-016-00251 |
| Market Analysis Reports |