Online Database of Chemicals from Around the World
| Tianjin Pharmacn Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharmacn.com | |||
 | +86 (22) 8372-6121 | |||
 | +86 (22) 2337-7103 | |||
 | marketing@pharmacn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2006 | ||||
| SinoPep-Allsino Biopharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinopep.com | |||
 | +86 (571) 8867-1330 +86 18167156872 | |||
 | +86 (571) 8629-8703 | |||
 | info@sinopep.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Cangzhou Senary Chemical S & T Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.senary.com | |||
 | +86 (317) 548-9300 | |||
 | +86 (317) 548-9300 | |||
 | sale01@senary.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Enantiotech Corporation Limited | China | |||
|---|---|---|---|---|
 | www.enantiotech.com | |||
 | +86 (760) 8528-2375 | |||
 | +86 (760) 8528-2626 | |||
 | marketing@enantiotech.net | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Ningbo NFC Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nfcchem.com | |||
 | +86 (574) 8799-1182 | |||
 | +86 (574) 8799-1185 | |||
 | sales@nfcchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Neosun Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neosunpharm.com | |||
 | +86 (21) 2095-6531 +86 18116226605 +86 18939712385 | |||
 | +86 (21) 2095-6532 | |||
 | neosunpharm@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Shilo Pharmachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shilopharma.com | |||
 | +86 15258872085 | |||
 | sales@shilopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Rochi Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rochipharma.com | |||
 | +86 (21) 3875-1876 +86 15000076078 | |||
 | +86 (21) 5027-5764 | |||
 | info@rochipharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Weifang Runzhong Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runzhongchem.com | |||
 | +86 (536) 786-8377 | |||
 | +86 (536) 7869677 | |||
 | sq@runzhongchem.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Zhejiang Zetian Fine Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zetchem.com | |||
 | +86 (571) 8732-2520 | |||
 | sales@zetchem.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shandong Dinghao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sddhpharm.com | |||
 | +86 (0531) 5856-5868 | |||
 | lily.zhou@sddhpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Jinan Hanyu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hanyuchem.com | |||
 | +86 (531) 6995-4981 | |||
 | sales@hanyuchem.com | |||
 | QQ Chat | |||
 | WeChat: dwyane-wang | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Sinbond Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinbondpharm.com | |||
 | +86 (531) 8703-8285 +86 13583181986 +44 (208) 242-5518 | |||
 | +86 (531) 8703-8285 | |||
 | cici1124@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| Classification | Organic raw materials >> Aryl compounds >> Anilines |
|---|---|
| Name | 4-Fluoro-2-methoxy-5-nitroaniline |
| Molecular Structure | 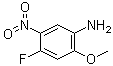 |
| Molecular Formula | C7H7FN2O3 |
| Molecular Weight | 186.14 |
| CAS Registry Number | 1075705-01-9 |
| EC Number | 806-172-0 |
| SMILES | COC1=CC(=C(C=C1N)[N+](=O)[O-])F |
| Solubility | Very slightly soluble (0.52 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.412±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 127 - 129 °C (Expl.) |
| Boiling point | 354.8±37.0 °C 760 mmHg (Calc.)* |
| Flash point | 168.4±26.5 °C (Calc.)* |
| Index of refraction | 1.578 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
4-Fluoro-2-methoxy-5-nitroaniline is an aromatic organic compound that belongs to the class of substituted anilines, featuring a fluorine atom at the 4-position, a methoxy group at the 2-position, and a nitro group at the 5-position of the benzene ring. Its discovery traces back to efforts in the mid-20th century to develop functionalized aniline derivatives as intermediates for dyes, agrochemicals, and pharmaceuticals. Functionalization of the aniline ring with electron-donating and electron-withdrawing groups, such as methoxy and nitro substituents respectively, has been a long-established strategy for tuning the reactivity and physical properties of these compounds. The preparation of 4-fluoro-2-methoxy-5-nitroaniline typically involves nucleophilic aromatic substitution reactions or electrophilic nitration strategies starting from fluorinated anisole derivatives. In one common approach, selective nitration of 4-fluoro-2-methoxyaniline or similar precursors under controlled conditions affords the target compound with high regioselectivity. The introduction of a nitro group is particularly significant as it not only modifies the electronic character of the molecule but also provides a versatile handle for further chemical transformations, such as reduction to an amine or participation in coupling reactions. Applications of 4-fluoro-2-methoxy-5-nitroaniline are primarily found in the fields of medicinal chemistry and materials science. It is frequently employed as an intermediate in the synthesis of more complex molecules, especially in the design of heterocyclic structures such as quinolines, pyridines, and benzodiazoles. The presence of both electron-donating and electron-withdrawing groups on the aromatic ring makes it a valuable building block for constructing compounds with specific electronic characteristics, which are essential in drug discovery programs targeting kinases, receptors, or enzymes. In addition to its role in medicinal chemistry, 4-fluoro-2-methoxy-5-nitroaniline has been investigated for applications in dye chemistry and material sciences. Substituted aniline derivatives such as this one are key components in the preparation of azo dyes and pigments, where the precise substitution pattern directly influences the absorption properties, colorfastness, and stability of the final materials. The fluorine atom enhances chemical resistance and modifies the hydrophobicity of derived dyes, while the methoxy group can improve solubility in organic solvents. The chemical also serves as a starting material for the synthesis of fluorinated benzene derivatives through further functionalization steps, including cross-coupling reactions such as Suzuki-Miyaura or Buchwald-Hartwig couplings. These transformations expand the utility of 4-fluoro-2-methoxy-5-nitroaniline in advanced organic synthesis, allowing the construction of structurally complex and diverse molecules required for modern applications in pharmaceuticals, agrochemicals, and organic electronics. Overall, 4-fluoro-2-methoxy-5-nitroaniline is a well-established, multifunctional intermediate that has found enduring use in synthetic chemistry, particularly where controlled reactivity and electronic modulation of aromatic systems are needed. Its discovery and utilization are rooted in classic organic chemistry strategies that continue to support innovation across multiple industries. References 2016. Osimertinib. Pharmaceutical Substances. 2022. Discovery of mobocertinib, a new irreversible tyrosine kinase inhibitor indicated for the treatment of non-small-cell lung cancer harboring EGFR exon 20 insertion mutations. Medicinal Chemistry Research, 31(10). DOI: 10.1007/s00044-022-02952-5 2023. Design, synthesis and biological evaluation of aminopyrimidine derivatives bearing dihydroquinoxalinone as novel EGFRL858R/T790M kinase inhibitors against non-small-cell lung cancer. Medicinal Chemistry Research, 32(6). DOI: 10.1007/s00044-023-03054-6 |
| Market Analysis Reports |