Online Database of Chemicals from Around the World
| Shanghai RC Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rcc.net.cn | |||
 | +86 (21) 5032-2175 5866-1250 ex 603 | |||
 | +86 (21) 5032-2176 / 5866-1251 | |||
 | sales@rcc.net.cn chad@rcc.net.cn | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Tyger Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.tygersci.com | |||
 | +1 (609) 434-0144 | |||
 | +1 (609) 434-0143 | |||
 | sales@tygersci.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2008 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| A.S. Chemical Laboratories Inc. | Canada | |||
|---|---|---|---|---|
 | www.aschemicals.com | |||
 | +1 (905) 669-7472 | |||
 | sales@aschemicals.com | |||
| Chemical manufacturer since 1997 | ||||
| Advanced Chemical Intermediates Ltd. | UK | |||
|---|---|---|---|---|
 | www.acints.com | |||
 | +44 (1840) 261-451 | |||
 | +44 (1840) 261-451 | |||
 | enquiries@acints.com | |||
| Chemical manufacturer | ||||
| AB Chem, Inc. | Canada | |||
|---|---|---|---|---|
 | www.abcheminc.com | |||
 | +1 (514) 685-8688 | |||
 | +1 (514) 685-8488 | |||
 | sales@abcheminc.com | |||
| Chemical manufacturer | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| CNH Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.cnhtechnologies.com | |||
 | +1 (781) 933-0362 | |||
 | +1 (781) 933-1839 | |||
 | info@cnhtechnologies.com | |||
| Chemical manufacturer | ||||
| Chess GmbH | Germany | |||
|---|---|---|---|---|
 | www.chess-chem.com | |||
 | +49 (621) 318-9794 | |||
 | +49 (621) 318-9691 | |||
 | sales@chess-chem.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Jintan Kangyuan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jtkychem.com | |||
 | +86 (519) 8256-6282 8256-6295 8269-9398 | |||
 | +86 (519) 8256-6281 / 8288-6169 | |||
 | sales@jtkychem.com | |||
| Chemical manufacturer since 1991 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | N-BOC-4-Hydroxypiperidine |
| Synonyms | N-(tert-Butoxycarbonyl)-4-hydroxypiperidine |
| Molecular Structure | 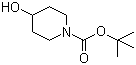 |
| Molecular Formula | C10H19NO3 |
| Molecular Weight | 201.26 |
| CAS Registry Number | 109384-19-2 |
| EC Number | 600-916-6 |
| SMILES | CC(C)(C)OC(=O)N1CCC(CC1)O |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 61 - 65 °C (Expl.) |
| Boiling point | 292.3±33.0 °C 760 mmHg (Calc.)* |
| Flash point | 130.6±25.4 °C (Calc.)* |
| Index of refraction | 1.496 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
N-BOC-4-Hydroxypiperidine is a Boc-protected 4-hydroxypiperidine featuring a piperidine ring with a tert-butoxycarbonyl (Boc) group on the nitrogen and a hydroxyl moiety at the 4-position. Its molecular formula is C10H19NO3, and it typically appears as a white or off-white crystalline powder, highly soluble in organic solvents such as methanol, ethanol, dichloromethane, and chloroform. The Boc protection enhances its chemical stability and handling. This compound is primarily employed as a versatile building block in organic synthesis, especially in pharmaceutical research and development. The Boc group serves as a temporary protective group for the amine, allowing selective reactions elsewhere. This facilitates multi-step syntheses of piperidine-containing bioactive molecules while maintaining control over functional group reactivity. Upon completion of downstream transformations, the Boc group can be removed under mild acidic conditions to release the free amine. In medicinal chemistry, N-BOC-4-Hydroxypiperidine is used in the synthesis of neurologically active agents, peptide analogs, and small-molecule drugs. It functions effectively in Mitsunobu etherification to create N-heterocyclic alkyl ethers. It also serves in the preparation of BTK inhibitor intermediates, and has been implicated in the assembly of compounds with activity at muscarinic receptors and mineralocorticoid receptors. The combination of the hydroxyl group and Boc protection provides branching points for further modification, including esterification, ether formation, and conjugation with other pharmacophores. The synthetic route often involves the reaction of 4-hydroxypiperidine with di-tert-butyl dicarbonate (Boc₂O) in the presence of base such as sodium bicarbonate, typically in dichloromethane or aqueous-organic mixtures at ambient temperature. The reaction proceeds with high yield and furnished N-BOC-4-Hydroxypiperidine as a crystalline solid. For industrial-scale production, a two-step approach starting from 4-piperidone includes reduction to 4-hydroxypiperidine followed by Boc protection. This process yields high purity product suitable as a pharmaceutical intermediate. As a reagent, N-BOC-4-Hydroxypiperidine is commercially available in high-purity grades (≥97 %), and stored at cool conditions. It is classified as an irritant, requiring standard protective equipment to avoid skin, eye, and respiratory exposure. Handling should be conducted in well-ventilated areas. In summary, N-BOC-4-Hydroxypiperidine is an important Boc-protected amino alcohol used extensively in pharmaceutical synthesis. Its dual functionality enables selective transformations, while its relatively simple preparation and commercial availability make it a staple intermediate for piperidine-based drug research. References 2024. Scalable deoxygenative alkynylation of alcohols via flow photochemistry. Communications Chemistry, 7(1). DOI: 10.1038/s42004-024-01363-4 2022. Discovery of small molecule inhibitors of Plasmodium falciparum apicoplast DNA polymerase. Journal of Enzyme Inhibition and Medicinal Chemistry, 37(1). DOI: 10.1080/14756366.2022.2070909 2018. HS-1371, a novel kinase inhibitor of RIP3-mediated necroptosis. Experimental & Molecular Medicine, 50(9). DOI: 10.1038/s12276-018-0152-8 |
| Market Analysis Reports |