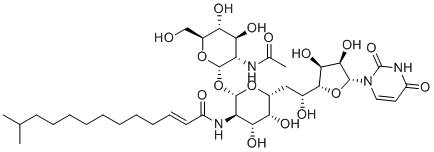
Tunicamycin is a nucleoside antibiotic complex produced by several strains of Streptomyces, particularly Streptomyces lysosuperificus. It was first isolated in the 1970s during a systematic screening for antimicrobial agents that inhibit bacterial cell wall synthesis. Structurally, tunicamycin is characterized by a uracil nucleoside moiety linked to a tunicamine sugar, bearing a fatty acid side chain. The compound exists as a mixture of homologs differing in the length of the fatty acid chain.
The primary mechanism of action of tunicamycin is the inhibition of N-acetylglucosamine-1-phosphate transferase, the enzyme responsible for catalyzing the first step in the biosynthesis of N-linked glycoproteins. Specifically, tunicamycin blocks the transfer of N-acetylglucosamine-1-phosphate to dolichol phosphate, thereby preventing the formation of dolichol-linked oligosaccharide intermediates. This inhibition disrupts protein glycosylation pathways in eukaryotic cells and interferes with peptidoglycan biosynthesis in certain bacterial species.
In cell biology and biochemical research, tunicamycin has become an essential tool for studying the role of N-linked glycosylation in protein folding, trafficking, and secretion. Its ability to selectively inhibit glycosylation without affecting other cellular processes has enabled detailed investigations into the functions of glycoproteins in various physiological and pathological contexts. In mammalian cells, exposure to tunicamycin induces endoplasmic reticulum stress by disrupting protein processing and activating the unfolded protein response.
The compound is also widely used to model diseases associated with defects in protein glycosylation. In experimental systems, tunicamycin treatment mimics the cellular stress conditions observed in disorders such as congenital disorders of glycosylation, enabling researchers to study molecular mechanisms and potential interventions. Furthermore, tunicamycin has been employed in cancer research to explore the dependency of tumor cells on glycoprotein biosynthesis and to evaluate the therapeutic potential of glycosylation inhibitors.
In microbiology, tunicamycin has been studied for its antibacterial activity against Gram-positive bacteria, including Bacillus subtilis and Staphylococcus aureus. Its effect is due to the inhibition of the early steps in the synthesis of bacterial cell wall precursors, leading to cell lysis. However, due to its cytotoxicity in eukaryotic cells, tunicamycin has not been developed for clinical use as an antimicrobial agent.
In addition to its role in protein glycosylation studies, tunicamycin has been applied in the analysis of lipid-linked oligosaccharide intermediates. Biochemical techniques utilizing tunicamycin have facilitated the identification and characterization of glycan assembly pathways and dolichol-phosphate-linked intermediates in various organisms. It has also been used to examine the fidelity of glycosylation and the quality control mechanisms within the secretory pathway.
Tunicamycin has further contributed to the field of developmental biology. In animal models, treatment with tunicamycin has revealed the essential role of N-linked glycosylation during embryogenesis, organ development, and cell differentiation. Its application has provided insights into the molecular consequences of glycosylation defects and the downstream effects on signaling pathways and gene expression.
Despite its usefulness in research, the use of tunicamycin requires careful handling due to its toxicity and stability profile. It is typically prepared as a solution in dimethyl sulfoxide or methanol and stored under low-temperature conditions to preserve its activity. Its potency and ability to induce rapid cellular responses necessitate precise dosage control in experimental designs.
In summary, tunicamycin represents a critical biochemical reagent with a well-defined mechanism of action that has enabled a wide range of studies in enzymology, glycobiology, microbiology, and disease modeling. Its contribution to the understanding of N-linked glycosylation and cellular stress responses continues to be of significant importance in biomedical research.
References
none
|



































 GHS06 Danger Details
GHS06 Danger Details