Online Database of Chemicals from Around the World
| Wuxi Orient Detergent Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinobromide.com | |||
 | +86 (510) 8758-1290 | |||
 | +86 (510) 8758-3694 | |||
 | suguangming@126.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Yancheng Longshen Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.longshenchem.com | |||
 | +86 (515) 8773-1896 +86 18261286666 | |||
 | +86 (515) 8865-2999 | |||
 | 2355756927@qq.com gaojunwyt@vip.163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Yick-Vic Chemicals and Pharmaceuticals (HK) Ltd. | Hong Kong | |||
|---|---|---|---|---|
 | www.yickvic.com | |||
 | +852 2541-2772 | |||
 | +852 2542-3444 | |||
 | yickvic@hkstar.com | |||
| Chemical distributor since 1982 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Anhui Weixiang New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ahwxchem.com | |||
 | +86 13195623283 | |||
 | export@ahwxchem.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Sanmenxia Aoke Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aokeby.com | |||
 | +86 (398) 383-4379 | |||
 | +86 (398) 383-0625 | |||
 | h99139@hotmail.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Halogenated aliphatic hydrocarbon |
|---|---|
| Name | Lauryl chloride |
| Synonyms | 1-Chlorododecane; Dodecyl chloride |
| Molecular Structure | 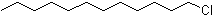 |
| Molecular Formula | C12H25Cl |
| Molecular Weight | 204.78 |
| CAS Registry Number | 112-52-7 |
| EC Number | 203-981-5 |
| SMILES | CCCCCCCCCCCCCl |
| Density | 0.9±0.1 g/cm3 Calc.*, 0.864 g/mL (Expl.) |
|---|---|
| Melting point | -9.3 °C (Expl.) |
| Boiling point | 260.4±3.0 °C 760 mmHg (Calc.)*, 260 °C (Expl.) |
| Flash point | 104.5±2.8 °C (Calc.)*, 130 °C (Expl.) |
| Solubility | water 0.0068 g/L (20 °C) (Expl.) |
| Index of refraction | 1.438 (Calc.)*, 1.442-1.444 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS07;GHS08;GHS09 Warning Details GHS07;GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H351-H400-H410 Details | ||||||||||||||||||||||||||||
| Safety Statements | P203-P264-P273-P280-P302+P352-P318-P321-P332+P317-P362+P364-P391-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 3082 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Lauryl chloride, also known as dodecyl chloride, is a straight-chain primary alkyl chloride with the molecular formula C12H25Cl. It is a colorless to pale yellow liquid with a characteristic odor, widely recognized as a reactive intermediate in organic synthesis and industrial chemistry. Lauryl chloride was first investigated in the early 20th century during studies of long-chain organohalogen compounds, as chemists sought to understand the chemical behavior of chlorinated fatty alcohol derivatives and their applications in surfactant and materials chemistry. Lauryl chloride is primarily produced by the chlorination of lauryl alcohol (dodecanol) using reagents such as thionyl chloride, phosphorus pentachloride, or other suitable halogenating agents. This transformation exemplifies a classic nucleophilic substitution reaction, where the hydroxyl group is replaced by a chlorine atom to form a more reactive alkyl halide. The compound can also be obtained via free radical chlorination of dodecane under controlled conditions, although this method requires precise regulation to favor the terminal chloride over other positional isomers. These synthetic approaches provide lauryl chloride in high purity, suitable for both laboratory and industrial applications. The reactivity of lauryl chloride is largely determined by the electrophilic carbon bonded to chlorine, which serves as an excellent leaving group in nucleophilic substitution reactions. This property allows lauryl chloride to act as an alkylating agent for a variety of nucleophiles, including alcohols, amines, thiols, and carboxylates, producing dodecyl ethers, amines, thioethers, and esters. As such, lauryl chloride is a key building block in the synthesis of surfactants, quaternary ammonium salts, and other long-chain derivatives, providing a hydrophobic C12 segment that imparts specific chemical and physical properties. In industrial applications, lauryl chloride is extensively used in the production of detergents, emulsifiers, and fabric softeners. It serves as a precursor for the manufacture of dodecyltrimethylammonium chloride and other quaternary ammonium compounds, which are valued for their antimicrobial properties and ability to reduce surface tension in aqueous formulations. Lauryl chloride derivatives are also employed in the synthesis of lubricants, corrosion inhibitors, and specialty chemicals, demonstrating versatility in both personal care and industrial chemistry sectors. The compound also plays an important role in mechanistic studies of organic reactions. As a primary chloride, lauryl chloride is an ideal substrate for exploring nucleophilic substitution (SN2) mechanisms, as the steric hindrance around the reactive site is minimal, facilitating direct bimolecular attack. Its behavior in various solvents and under differing nucleophilic conditions has contributed to a deeper understanding of structure–reactivity relationships in alkyl halides and the effects of chain length on reaction kinetics. Physically, lauryl chloride is immiscible with water, soluble in organic solvents such as ether, chloroform, and alcohols, and has a relatively high boiling point due to van der Waals interactions among the long hydrocarbon chains. These properties are significant for handling, reaction design, and purification processes. Safety considerations are also critical; lauryl chloride is corrosive and can cause skin, eye, and respiratory irritation. Proper ventilation, personal protective equipment, and adherence to safety protocols are necessary to mitigate exposure risks. Environmental management is essential due to its potential persistence and toxicity to aquatic life. The discovery and application of lauryl chloride highlight its significance as a versatile organohalogen compound. Its role as a reactive intermediate in synthetic organic chemistry and its widespread use in industrial formulations underscore its continued relevance, making it a fundamental reagent for the production of surfactants, quaternary ammonium compounds, and other long-chain chemical derivatives. References 2020. Functionalized imidazolium-based ionic liquids: biological activity evaluation, toxicity screening, spectroscopic, and molecular docking studies. Medicinal Chemistry Research, 29(10). DOI: 10.1007/s00044-020-02631-3 2019. Identification of Anionic and Nonionic Surfactant and Recalcitrants Compounds in Commercial Laundry Wastewater by GC-MS Analysis After Anaerobic Fluidized Bed Reactor Treatment. Water, Air, & Soil Pollution, 230(12). DOI: 10.1007/s11270-019-4357-9 2019. Investigating the effect of [C8Py][Cl] and [C18Py][Cl] ionic liquids on the water/oil interfacial tension by considering Taguchi method. Journal of Petroleum Exploration and Production Technology, 9(4). DOI: 10.1007/s13202-019-0688-8 |
| Market Analysis Reports |