Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Biochemical >> Condensing agent |
|---|---|
| Name | 4,5-Dicyanoimidazole |
| Synonyms | 1H-Imidazole-4,5-dicarbonitrile; DCI |
| Molecular Structure | 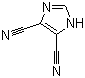 |
| Molecular Formula | C5H2N4 |
| Molecular Weight | 118.09 |
| CAS Registry Number | 1122-28-7 |
| EC Number | 214-344-6 |
| SMILES | C1=NC(=C(N1)C#N)C#N |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 168-175 °C (Expl.) |
| Index of Refraction | 1.584, Calc.* |
| Boiling Point | 569.3±35.0 °C (760 mmHg), Calc.* |
| Flash Point | 170.2±11.1 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H318-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
4,5-Dicyanoimidazole is a highly reactive heterocyclic compound with a structure consisting of an imidazole ring substituted at positions 4 and 5 with cyano groups. This arrangement of the cyano groups significantly enhances the electrophilic nature of the compound, making it a versatile reagent in organic synthesis and chemical reactions. The compound is mainly utilized as a nucleophilic catalyst or in the preparation of more complex organic molecules, such as heterocyclic compounds, pharmaceuticals, and agrochemicals. The discovery of 4,5-dicyanoimidazole can be traced to the growing interest in the chemistry of imidazole derivatives in the mid-20th century. Imidazole itself, with its nitrogen-containing five-membered ring, had already been established as a valuable scaffold for a variety of chemical and biological activities. The introduction of cyano groups at positions 4 and 5 was found to dramatically alter the reactivity of the imidazole ring, imparting unique properties that made 4,5-dicyanoimidazole an important compound in the development of synthetic methodologies and catalysis. One of the primary applications of 4,5-dicyanoimidazole is in organic synthesis, where it is employed as a catalyst or a reagent for the formation of carbon-carbon bonds. Its ability to facilitate nucleophilic substitution reactions and its electron-withdrawing cyano groups make it an excellent candidate for promoting various transformations, such as the formation of imidazole-based derivatives. It has been used in the synthesis of polycyclic aromatic compounds, which have applications in materials science, and in the creation of advanced intermediates for pharmaceutical synthesis. 4,5-Dicyanoimidazole is also used in the synthesis of more complex heterocyclic compounds. Its reactivity allows it to participate in cyclization reactions, where it serves as both a reactant and a catalyst. These reactions are critical in the formation of structurally diverse compounds that find uses in drug development, agrochemical research, and the production of functional materials. Its application in the synthesis of biologically active heterocycles has led to its incorporation into the design of novel therapeutic agents. In addition to its use in organic synthesis, 4,5-dicyanoimidazole has been explored for its potential role in catalysis. It has been found to be a potent catalyst in a range of reactions, such as nucleophilic substitution, cycloaddition, and oxidation reactions. Its ability to activate certain functional groups, particularly carbon-carbon bonds, makes it useful in a variety of catalytic processes. This has significant implications for the development of green chemistry techniques, where catalysts are used to reduce the environmental impact of chemical reactions. Moreover, 4,5-dicyanoimidazole has applications in materials science, particularly in the development of molecular electronics and as a building block for the creation of conductive materials. Due to the electron-withdrawing nature of the cyano groups, it has been explored as part of donor-acceptor systems in organic electronics, where it can serve to modulate the electronic properties of materials used in flexible electronics, organic light-emitting diodes (OLEDs), and organic photovoltaics (OPVs). In conclusion, 4,5-dicyanoimidazole is a highly versatile and reactive compound that has found a broad range of applications in organic synthesis, catalysis, and materials science. Its ability to participate in a variety of chemical reactions and facilitate the formation of heterocyclic compounds has made it an essential tool in the development of new chemical processes and materials. Its potential for use in catalysis and molecular electronics further underscores its importance in modern chemistry and technology. References 1998. Efficient activation of nucleoside phosphoramidites with 4,5-dicyanoimidazole during oligonucleotide synthesis. Nucleic Acids Research. DOI: 10.1093/nar/26.4.1046 2017. Synthesis of new substituted 5-amino-1H-imidazole-4-carbonitriles. Russian Journal of General Chemistry. DOI: 10.1134/s1070363217100334 2019. Hydrogen-bonded supramolecular metal-imidazolate frameworks: gas sorption, magnetic and UV/Vis spectroscopic properties. Journal of Inclusion Phenomena and Macrocyclic Chemistry. DOI: 10.1007/s10847-019-00926-6 |
| Market Analysis Reports |