Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Ningbo Fengrui Fine Chemical Ltd. | China | |||
|---|---|---|---|---|
 | www.fengruichem.com | |||
 | +86 (574) 8772-0208 | |||
 | fr006@fengruichem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hubei Teyer Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hubeiteyer.com | |||
 | +86 (728) 533-2622 | |||
 | info@hubeiteyer.com | |||
| Chemical manufacturer since 2002 | ||||
| Classification | Biochemical >> Inhibitor >> Immune inhibitor |
|---|---|
| Name | Mycophenolate mofetil |
| Synonyms | 2-Morpholin-4-ylethyl (E)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1H-isobenzofuran-5-yl)-4-methyl-hex-4-enoate |
| Molecular Structure | 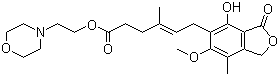 |
| Molecular Formula | C23H31NO7 |
| Molecular Weight | 433.50 |
| CAS Registry Number | 115007-34-6 |
| EC Number | 680-628-5 |
| SMILES | CC1=C2COC(=O)C2=C(C(=C1OC)C/C=C(C)/CCC(=O)OCCN3CCOCC3)O |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.557, Calc.* |
| Boiling Point | 637.6±55.0 °C (760 mmHg), Calc.* |
| Flash Point | 339.4±31.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS07;GHS08;GHS09 Danger Details GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H341-H360-H372——H400-H410 Details | ||||||||||||||||||||||||
| Safety Statements | P203-P260-P264-P270-P273-P280-P301+P317-P318-P319-P330-P391-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
|
Mycophenolate mofetil is a well-established immunosuppressive drug that serves as a prodrug of mycophenolic acid. It was developed to improve the pharmacokinetic properties of its active form, particularly its oral bioavailability and gastrointestinal tolerability. Mycophenolic acid, the active metabolite, was originally isolated in 1893 from *Penicillium glaucum*, but it was not until the late 20th century that its clinical potential as an immunosuppressant was fully realized. The mofetil ester of mycophenolic acid was introduced in the early 1990s to address limitations of the parent compound, and mycophenolate mofetil received regulatory approval for clinical use in organ transplantation shortly thereafter. The primary mechanism of action of mycophenolate mofetil is the inhibition of inosine monophosphate dehydrogenase (IMPDH), a key enzyme in the de novo synthesis of guanosine nucleotides. This pathway is particularly important for the proliferation of T and B lymphocytes, which rely on it more heavily than other cell types. As such, mycophenolate mofetil exerts selective antiproliferative effects on lymphocytes, contributing to its immunosuppressive properties without broadly impairing other rapidly dividing cells. Mycophenolate mofetil is widely used in clinical practice for the prevention of organ rejection in renal, cardiac, and hepatic transplant recipients. It is commonly administered as part of a combination regimen with calcineurin inhibitors and corticosteroids. Its use has been associated with improved graft survival and reduced incidence of acute rejection episodes. In addition to transplantation, mycophenolate mofetil is also employed off-label or under specific guidelines in the treatment of various autoimmune diseases, including lupus nephritis, autoimmune hepatitis, and certain vasculitides, based on its immunomodulatory effects. The drug is administered orally, and its ester form is rapidly hydrolyzed in vivo to yield the active mycophenolic acid. Mycophenolate mofetil has favorable absorption characteristics compared to mycophenolic acid itself, making it suitable for long-term use. Its pharmacokinetics are well-characterized, with enterohepatic recirculation contributing to a secondary peak in plasma concentration after dosing. Safety considerations for mycophenolate mofetil include its known teratogenicity and increased risk of infections due to immunosuppression. It is contraindicated in pregnancy, and patients on therapy are advised to use effective contraception. Common adverse effects include gastrointestinal disturbances such as diarrhea, nausea, and abdominal pain, as well as hematologic effects like leukopenia and anemia. Mycophenolate mofetil has become an essential agent in modern immunosuppressive therapy, with a well-documented efficacy and safety profile. Its introduction marked a significant advancement in the management of transplant recipients and patients with autoimmune diseases, helping to achieve better clinical outcomes through targeted immunosuppression. References 2007. Kidney Transplantation With Sirolimus and Mycophenolate Mofetil-Based Immunosuppression: 5-Year Results of a Randomized Prospective Trial Compared to Calcineurin Inhibitor Drugs. Transplantation, 83(7). DOI: 10.1097/01.tp.0000258586.52777.4c 2007. Mycophenolate mofetil versus azathioprine for prevention of chronic allograft dysfunction in renal transplantation: the MYSS follow-up randomized, controlled clinical trial. Journal of the American Society of Nephrology, 18(6). DOI: 10.1681/asn.2006101153 2004. Mycophenolate mofetil. Acta Crystallographica Section E Structure Reports Online, 60(11). DOI: 10.1107/s1600536804027539 |
| Market Analysis Reports |