Online Database of Chemicals from Around the World
| Hangzhou StarShine Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.starshinepharm.com | |||
 | +86 (571) 8512-3681 +86 13777804878 | |||
 | +86 (571) 8512-2157 | |||
 | sales@starshinepharm.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beijing Team Academy of Pharmaceutical Sciences Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.thpharms.com | |||
 | +86 (10) 6818-0921 | |||
 | +86 (10) 6818-7090 | |||
 | kbryant8@126.com kbryant2010@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Molcan Corporation | USA | |||
|---|---|---|---|---|
 | www.molcan.com | |||
 | +1 (905) 731-5537 | |||
 | +1 (905) 248-3843 | |||
 | info@molcan.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Shilo Pharmachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shilopharma.com | |||
 | +86 15258872085 | |||
 | sales@shilopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Classification | API >> Inhibitor drug |
|---|---|
| Name | Tetrabenzyl-voglibose |
| Synonyms | TBV; 2-[[(1S,2S,3R,4S,5S)-5-hydroxy-2,3,4-tris(phenylmethoxy)-5-(phenylmethoxymethyl)cyclohexyl]amino]propane-1,3-diol |
| Molecular Structure | 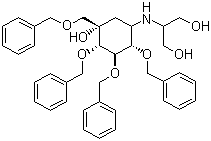 |
| Molecular Formula | C38H45NO7 |
| Molecular Weight | 627.77 |
| CAS Registry Number | 115250-39-0 |
| SMILES | C1[C@@H]([C@@H]([C@H]([C@@H]([C@]1(COCC2=CC=CC=C2)O)OCC3=CC=CC=C3)OCC4=CC=CC=C4)OCC5=CC=CC=C5)NC(CO)CO |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.630, Calc.* |
| Boiling Point | 784.1±60.0 °C (760 mmHg), Calc.* |
| Flash Point | 428.0±32.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P351-P302+P352 Details |
| SDS | Available |
|
Tetrabenzyl-voglibose is a synthetic derivative of voglibose, a potent alpha-glucosidase inhibitor. The discovery of tetrabenzyl-voglibose arose from efforts to modify voglibose for enhanced pharmacological properties and improved synthesis pathways. Voglibose itself was developed in the 1980s in Japan as an oral anti-diabetic drug to manage postprandial hyperglycemia by delaying carbohydrate absorption. This inhibition mechanism targets enzymes such as sucrase, maltase, and isomaltase, reducing glucose uptake and controlling blood sugar levels in diabetic patients. However, voglibose’s structural characteristics, including multiple hydroxyl groups, posed challenges for certain chemical manipulations and derivatizations. The introduction of benzyl groups to voglibose resulted in tetrabenzyl-voglibose, which plays a significant role in synthetic organic chemistry as a protected intermediate. By replacing hydroxyl groups with benzyl groups, the molecule becomes more amenable to further synthetic transformations. This modification facilitates regioselective functionalization and the development of voglibose analogs with potential therapeutic applications. In particular, tetrabenzyl-voglibose enables chemists to create derivatives that may exhibit enhanced bioavailability, stability, or selectivity toward specific enzymes. One notable application of tetrabenzyl-voglibose is in the synthesis of voglibose and its analogs via selective deprotection strategies. The benzyl groups can be removed under controlled conditions, yielding voglibose or modified analogs with minimal byproducts. This approach streamlines the synthesis process, ensuring higher yields and purity. Moreover, the benzylated form of voglibose offers insight into the structure-activity relationships of alpha-glucosidase inhibitors, aiding in the design of novel anti-diabetic agents. In pharmaceutical research, tetrabenzyl-voglibose serves as a scaffold for creating new therapeutic candidates aimed at improving glycemic control. By modifying the tetrabenzyl structure, researchers can explore variations that may optimize drug delivery, minimize side effects, or increase the duration of action. This versatility makes tetrabenzyl-voglibose valuable not only in diabetes management but also in broader metabolic disorder research. Overall, tetrabenzyl-voglibose represents a crucial intermediate in medicinal chemistry, enabling advancements in the development of carbohydrate metabolism modulators. Its discovery highlights the importance of strategic chemical modifications in enhancing the utility of existing drugs and facilitating the synthesis of novel therapeutic agents. References none |
| Market Analysis Reports |