Online Database of Chemicals from Around the World
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound |
|---|---|
| Name | 3-Chloro-4-iodo-2-pyridinamine |
| Synonyms | (3-Chloro-4-iodopyridin-2-yl)amine |
| Molecular Structure | 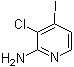 |
| Molecular Formula | C5H4ClIN2 |
| Molecular Weight | 254.46 |
| CAS Registry Number | 1152617-24-7 |
| SMILES | C1=CN=C(C(=C1I)Cl)N |
| Solubility | Slightly soluble (1.1 g/L) (25 °C), Calc.* |
|---|---|
| Density | 2.139±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 308.9±42.0 °C 760 mmHg (Calc.)* |
| Flash point | 140.6±27.9 °C (Calc.)* |
| Index of refraction | 1.708 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319 Details |
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details |
| SDS | Available |
|
3-Chloro-4-iodo-2-pyridinamine is an organic compound containing a pyridine ring with two halogen substituents and an amine group. Specifically, the compound consists of a pyridine core with a chlorine atom at the 3-position, an iodine atom at the 4-position, and an amine group (-NH2) at the 2-position. Pyridine derivatives like this one have been widely studied for their potential applications in various chemical and biological fields. The synthesis of 3-chloro-4-iodo-2-pyridinamine generally involves halogenation reactions on a pyridine ring. In this case, the process would require selective chlorination and iodination of the pyridine core. The amine group, which is already in the 2-position, is often introduced early in the synthesis through nitration followed by reduction or via other direct amination reactions. Halogenation reactions are typically carried out under controlled conditions, using reagents such as chlorine gas or iodine in the presence of a suitable solvent and catalyst, to ensure the correct positioning of the halogens on the pyridine ring. This compound has significant potential for use in medicinal chemistry and materials science due to its unique structure and functional groups. The amine group, which is a common functional group in pharmaceuticals, can participate in hydrogen bonding and other interactions with biological targets, potentially making the compound bioactive. Additionally, the halogen atoms, particularly iodine, can influence the electronic properties of the molecule and may make it useful in applications like imaging or as a precursor to other more complex compounds. One notable application of halogenated pyridines, such as 3-chloro-4-iodo-2-pyridinamine, is in the development of pharmaceutical agents. Pyridine derivatives are often found in drug molecules that target enzymes, receptors, or other biomolecules. The halogen substituents can modulate the compound's pharmacokinetic properties, such as its solubility, stability, and ability to cross biological membranes. Specifically, the iodine atom may enhance the molecule's lipophilicity, aiding in the compound's ability to penetrate cell membranes. Moreover, halogenated pyridines are frequently used in the synthesis of more complex molecules, such as ligands for metal coordination complexes, which are important in areas like catalysis and material science. The halogen atoms can provide additional reactivity and allow the compound to be further modified or incorporated into larger systems with diverse applications. In addition to medicinal chemistry, 3-chloro-4-iodo-2-pyridinamine may have applications in agrochemicals, such as herbicides or pesticides. Pyridine-based compounds are known for their biological activity, and halogenated derivatives have been explored for their potential to interfere with biological processes in pests or pathogens. The ability of this compound to act as a precursor in the synthesis of more complex agrochemical agents could make it valuable in agricultural chemistry. In conclusion, 3-chloro-4-iodo-2-pyridinamine is a versatile compound with applications in drug discovery, materials science, and potentially agrochemicals. Its halogenated pyridine structure, combined with the amine group, makes it an important intermediate for the synthesis of more complex bioactive molecules and coordination complexes. Further research into its chemical reactivity and biological activity could lead to novel applications in various fields. |
| Market Analysis Reports |