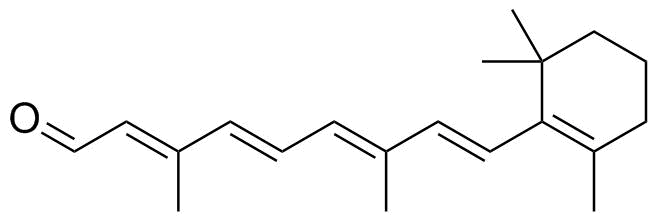
trans-Retinal is a naturally occurring aldehyde form of vitamin A (retinol) and plays a central role in vision and retinoid biochemistry. It consists of a conjugated polyene chain terminating in an aldehyde group (–CHO), and it exists predominantly in the all-trans configuration under physiological conditions. This extended conjugated system is responsible for its light-absorbing properties, which are essential for visual phototransduction.
The structure of trans-retinal is derived from beta-carotene metabolism. In animals, vitamin A (retinol) is oxidized to retinal through enzymatic action. The molecule contains a beta-ionone ring connected to an isoprenoid side chain with four conjugated double bonds, ending in an aldehyde functional group. The all-trans configuration refers to the geometry of the double bonds along the chain, which keeps the molecule in an extended, linear form that is chemically stable in the absence of light-induced isomerization.
The discovery of retinal and its role in vision is closely linked to early studies of visual pigments in the 20th century. Researchers investigating the chemistry of the retina identified a light-sensitive chromophore bound to the protein opsin, forming rhodopsin in rod cells. It was later determined that 11-cis-retinal, a geometric isomer of retinal, is the actual light-absorbing form in rhodopsin. Upon absorption of a photon, 11-cis-retinal is converted to all-trans-retinal, triggering a conformational change in opsin that initiates the visual signaling cascade. trans-Retinal is thus a key intermediate in the regeneration cycle of visual pigments.
In the visual cycle, all-trans-retinal is produced after photoactivation of rhodopsin. It is then released from opsin and reduced to all-trans-retinol by retinal dehydrogenases. This retinol is transported to the retinal pigment epithelium, where it is enzymatically converted back into 11-cis-retinal through a series of isomerization and oxidation steps. The regenerated 11-cis-retinal is then returned to photoreceptor cells to reconstitute rhodopsin, allowing continuous light sensitivity.
Beyond vision, retinal and its derivatives are important signaling molecules in biological systems. Retinoids, including retinoic acid (oxidized form of retinal), regulate gene expression during embryonic development, cell differentiation, and tissue maintenance. Retinal serves as a metabolic intermediate between retinol and retinoic acid, linking dietary vitamin A intake to both visual and gene regulatory functions.
trans-Retinal is chemically reactive due to its aldehyde group and conjugated double bond system. It can undergo isomerization, oxidation, and Schiff base formation with amino groups in proteins. In the visual system, it forms a reversible Schiff base linkage with a lysine residue in opsin, which is essential for chromophore binding and photoreceptor function.
Historically, the identification of retinal was a major milestone in understanding the molecular basis of vision. It helped establish the concept that light detection in biological systems is mediated by specific chemical transformations of chromophores embedded in proteins. This discovery contributed significantly to the fields of biochemistry, biophysics, and sensory biology.
Overall, trans-retinal is a biologically essential aldehyde form of vitamin A that functions as a key intermediate in the visual cycle. Its light-sensitive conjugated structure enables photon detection in the retina, while its reversible isomerization and metabolism support continuous visual function and broader roles in cellular signaling and development.
References
2026. Conformation-selective detection of residues in solid proteins under magic-angle-spinning. Journal of Biomolecular NMR.
DOI: 10.1007/s10858-025-00482-x
2026. Machine learning-assisted SERS detection of pyrethroid pesticides in edible fungi using a magnetic nanosensor. Microchimica Acta.
DOI: 10.1007/s00604-025-07832-4
2026. Cross-species optimization of nuclei isolation in ten plant species. Plant Methods.
DOI: 10.1186/s13007-025-01491-5
|



































 GHS07;GHS08 Danger Details
GHS07;GHS08 Danger Details