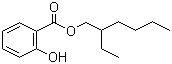
2-Ethylhexyl salicylate, commonly known as octyl salicylate, is an organic compound primarily used in the formulation of sunscreens and other cosmetic products. Its chemical formula is C13H18O3, and it is classified as an ester derived from salicylic acid and 2-ethylhexanol. The compound was first synthesized in the mid-20th century during a period of heightened interest in developing effective ultraviolet (UV) filters for skin protection. Researchers aimed to create substances that could effectively absorb UV radiation, particularly UVB rays, which are responsible for causing sunburn and contributing to skin cancer.
The discovery of 2-ethylhexyl salicylate marked a significant advancement in the cosmetic industry, providing formulators with a chemical that exhibits broad-spectrum UV absorption properties. This compound absorbs UV radiation in the UVB range (280-320 nm), helping to protect the skin from the harmful effects of sun exposure. As a result, it became a widely utilized ingredient in various sunscreen formulations, often combined with other UV filters to enhance overall effectiveness. In addition to its primary role as a UV filter, 2-ethylhexyl salicylate also serves as a solvent and a skin conditioning agent, improving the texture and application of cosmetic products.
The typical concentration of 2-ethylhexyl salicylate in sunscreen formulations ranges from 5% to 10%. Its compatibility with other sunscreen agents, such as avobenzone and octocrylene, enables formulators to create broad-spectrum sunscreens that protect against both UVA and UVB rays. The compound is known for its stability under sunlight, making it particularly suitable for outdoor use.
In addition to sunscreens, 2-ethylhexyl salicylate is also found in other personal care products, including moisturizers, lip balms, and makeup. Its ability to enhance the feel of these products makes it a versatile ingredient in the cosmetic industry.
Despite its widespread use, concerns have been raised regarding the potential health effects of certain sunscreen ingredients, including 2-ethylhexyl salicylate. Regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Commission have conducted safety assessments to evaluate its safety in cosmetic formulations. These evaluations are crucial to ensuring that the benefits of UV protection do not come at the expense of consumer safety.
As consumer preferences shift toward natural and organic products, the cosmetic industry is exploring alternative UV filters. However, 2-ethylhexyl salicylate continues to be a popular choice due to its proven efficacy and stability. Ongoing research aims to understand better the long-term effects of this compound on human health and the environment, ensuring that it remains a safe option for consumers.
In summary, 2-ethylhexyl salicylate is a significant chemical compound in the formulation of sunscreens and cosmetics, recognized for its UV filtering properties and versatility in personal care products. Its discovery in the mid-20th century has contributed to advancements in skin protection, although ongoing safety assessments are necessary to ensure its responsible use in the industry.
References
2024. New metabolites of 2-ethylhexyl salicylate in human urine after simulated real-life dermal sunscreen application. Toxicology Letters.
DOI: 10.1016/j.toxlet.2024.07.912
2024. Comprehensive assessment of the UV-filter 2-ethylhexyl salicylate and its phase I/II metabolites in urine by extended enzymatic hydrolysis and on-line SPE LC-MS/MS. Talanta.
DOI: 10.1016/j.talanta.2024.126223
2024. Food additive salicylates inhibit human and rat placental 3β-hydroxysteroid dehydrogenase: 3D-QSAR and in silico analysis. Chemico-Biological Interactions.
DOI: 10.1016/j.cbi.2024.111203
|












































 GHS07;GHS09 Warning Details
GHS07;GHS09 Warning Details