Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Chemical reagent >> Organic reagent >> Phenols |
|---|---|
| Name | 2-Cyclohexylphenol |
| Molecular Structure | 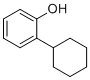 |
| Molecular Formula | C12H16O |
| Molecular Weight | 176.25 |
| CAS Registry Number | 119-42-6 |
| EC Number | 204-322-4 |
| SMILES | C1CCC(CC1)C2=CC=CC=C2O |
| Density | 1.0±0.1 g/cm3, Calc.* |
|---|---|
| Melting oint | 56 °C |
| Index of Refraction | 1.552, Calc.* |
| Boiling Point | 238.5±9.0 °C (760 mmHg), Calc.* |
| Flash Point | 113.8±8.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H315-H318-H335-H400-H410-H411 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P271-P273-P280-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P319-P321-P332+P317-P362+P364-P363-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
2-Cyclohexylphenol is an organic compound known for its distinctive structure, featuring a cyclohexyl group attached to the second position of a phenolic ring. With a molecular formula of C12H16O, this compound falls under the class of alkylphenols, which are valued for their chemical versatility and unique properties. The discovery and synthesis of 2-cyclohexylphenol emerged from the early exploration of phenolic derivatives, with researchers initially identifying it as a useful intermediate in organic synthesis. The addition of the cyclohexyl group to phenol imparts both structural stability and lipophilicity, enabling diverse applications across several fields. One of the primary uses of 2-cyclohexylphenol is in the development of specialty polymers. Its phenolic structure enhances the thermal and chemical stability of polymer products, making it a valuable additive for improving the performance of various plastic materials, particularly those used in high-temperature applications. The compound also finds application in resin formulations, where its presence contributes to the durability and flexibility of the end product. In addition to its role in materials science, 2-cyclohexylphenol has significant applications in the formulation of surfactants and detergents. The compound’s lipophilic properties, owing to the cyclohexyl group, improve the solubilizing ability of surfactant blends, enhancing their efficacy in removing oils and greases. This makes it an important ingredient in industrial cleaning agents as well as certain household products that require strong yet non-toxic cleaning action. In the realm of pharmaceuticals, 2-cyclohexylphenol has been studied for its potential bioactivity. Preliminary studies suggest that its phenolic structure, combined with the hydrophobic cyclohexyl substituent, may allow it to interact with lipid membranes, potentially leading to antifungal or antibacterial effects. While not widely used as a direct therapeutic agent, these properties have prompted research into its role as a chemical precursor for developing bioactive derivatives. Overall, 2-cyclohexylphenol represents a valuable chemical intermediate with applications in polymers, surfactants, and pharmaceutical research. Its unique structural features make it suitable for enhancing the properties of materials and formulations, and ongoing studies continue to reveal its versatility across industries. As a compound bridging the gap between synthetic chemistry and practical applications, 2-cyclohexylphenol holds a noteworthy place in modern chemical processes. References Rodrigo Amandi, Katherine Scovell, Peter Licence, Tobias J. Lotz and Martyn Poliakoff. The synthesis of o-cyclohexylphenol in supercritical carbon dioxide: towards a continuous two-step reaction, Green Chem., 2007, 9, 797. |
| Market Analysis Reports |