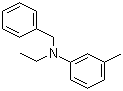
Ethylbenzyltoluidine, also known as 4-ethyl-4'-methylaminobenzophenone, is an organic compound with the chemical formula C16H17N. It belongs to the class of chemical substances known as arylamines and is characterized by the presence of an ethyl group, a benzyl group, and a toluidine moiety. The compound's discovery is part of the broader exploration of arylamines and their derivatives, which began in the late 19th and early 20th centuries as chemists sought to develop new compounds for various applications, including dyes, pharmaceuticals, and agrochemicals.
The synthesis of ethylbenzyltoluidine involves the reaction of ethylbenzene and toluidine, typically using traditional organic synthesis techniques such as amination or Friedel-Crafts alkylation. This compound was first reported in the mid-20th century, at a time when the demand for novel organic compounds in both the industrial and pharmaceutical sectors was rapidly increasing. Ethylbenzyltoluidine gained attention for its unique chemical structure and potential applications.
One of the primary applications of ethylbenzyltoluidine is in the field of organic synthesis as an intermediate for producing other chemicals. Its structure allows for further functionalization, enabling the creation of more complex molecules used in various industries. This compound is also employed in the manufacture of dyes and pigments, particularly in the textile industry, where it contributes to the coloration of fabrics and other materials.
In addition to its use in dye production, ethylbenzyltoluidine has been explored for its potential applications in pharmaceuticals. Research has indicated that arylamines, including this compound, may exhibit biological activity, making them candidates for further investigation as drug precursors or active pharmaceutical ingredients. The presence of the ethyl and toluidine groups in ethylbenzyltoluidine may contribute to its potential pharmacological properties, although more research is needed to fully understand its effects and applications in this area.
The safety and environmental impact of ethylbenzyltoluidine are also critical considerations in its application. As with many arylamines, concerns have been raised regarding their potential toxicity and carcinogenicity. Regulatory agencies, such as the U.S. Environmental Protection Agency (EPA) and the European Chemicals Agency (ECHA), evaluate the safety of chemical substances like ethylbenzyltoluidine to ensure that they are used responsibly in industrial and consumer applications. It is essential for manufacturers to adhere to safety guidelines and conduct thorough assessments of the compounds they use.
As industries increasingly shift towards sustainable and environmentally friendly practices, the role of compounds like ethylbenzyltoluidine may evolve. Researchers are exploring greener synthesis methods and alternative compounds that can fulfill similar roles without adverse effects on human health or the environment.
In summary, ethylbenzyltoluidine is an arylamine compound with significant potential in organic synthesis and industrial applications, particularly in dye production and pharmaceuticals. Its discovery and subsequent utilization reflect the ongoing need for innovative chemical compounds in various sectors. Continued research into its properties and applications will help to ensure that it can be used safely and effectively in the future.
References
1996. Use of incremental models to estimate the retention indexes of aromatic compounds. Chromatographia.
DOI: 10.1007/bf02292946
1998. Einstufung wassergefährdender Stöffe. Bundesgesundheitsblatt.
DOI: 10.1007/bf03044070
|



























 GHS06 Danger Details
GHS06 Danger Details