Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | API >> Hormone and endocrine-regulating drugs >> Gonadotropin |
|---|---|
| Name | Cetrorelix |
| Synonyms | (2S)-1-[(2S)-2-[[(2S)-2-[[(2R)-2-[[(2S)-2-[[(2S)-2-[[(2R)-2-[[(2R)-2-[[(2R)-2-acetamido-3-naphthalen-2-ylpropanoyl]amino]-3-(4-chlorophenyl)propanoyl]amino]-3-pyridin-3-ylpropanoyl]amino]-3-hydroxypropanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-5-(carbamoylamino)pentanoyl]amino]-4-methylpentanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]-N-[(2R)-1-amino-1-oxopropan-2-yl]pyrrolidine-2-carboxamide |
| Molecular Structure | 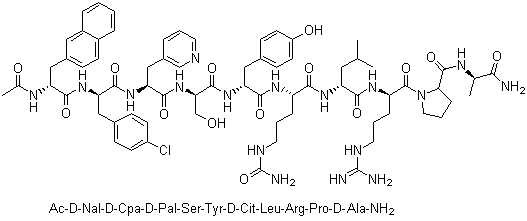 |
| Molecular Formula | C70H92ClN17O14 |
| Molecular Weight | 1431.04 |
| Protein Sequence | XXXSYXLRPA |
| CAS Registry Number | 120287-85-6 |
| EC Number | 686-384-6 |
| SMILES | C[C@H](C(=O)N)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CCCNC(=O)N)NC(=O)[C@H](CC2=CC=C(C=C2)O)NC(=O)[C@H](CO)NC(=O)[C@@H](CC3=CN=CC=C3)NC(=O)[C@@H](CC4=CC=C(C=C4)Cl)NC(=O)[C@@H](CC5=CC6=CC=CC=C6C=C5)NC(=O)C |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Index of refraction | 1.668 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS08 Danger Details GHS08 Danger Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H360-H360F Details | ||||||||||||
| Safety Statements | P203-P280-P318-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
|
HCetrorelix is a synthetic decapeptide that functions as a gonadotropin-releasing hormone (GnRH) receptor antagonist. It was developed to competitively inhibit the binding of endogenous GnRH to its receptors in the anterior pituitary, thereby suppressing the secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH). This mechanism allows for controlled ovarian stimulation protocols in assisted reproductive technology and is also used in managing hormone-sensitive conditions such as prostate cancer. The decapeptide sequence of cetrorelix has been optimized to provide high receptor affinity, enhanced stability against enzymatic degradation, and predictable pharmacokinetic properties. The discovery of cetrorelix arose from the need for reversible and controllable suppression of gonadotropin release, improving on previous strategies that used GnRH agonists. Unlike agonists, which initially stimulate LH and FSH release before desensitizing the pituitary, cetrorelix produces a rapid and reversible suppression of gonadotropin secretion without the initial hormone surge. This property is particularly valuable in in vitro fertilization protocols, where preventing premature ovulation is critical for successful oocyte retrieval. Cetrorelix is administered subcutaneously, and its pharmacodynamic profile shows rapid onset of action, with suppression of LH and FSH occurring within hours of injection. Its use has been extensively studied in women undergoing controlled ovarian hyperstimulation, demonstrating efficacy in preventing premature LH surges while maintaining follicular development. Additionally, cetrorelix has been investigated in men with prostate cancer, where its ability to suppress testosterone via LH inhibition offers a therapeutic approach complementary to other hormone therapies. Research into cetrorelix also highlights its safety and tolerability profile, with adverse effects generally mild and related to injection-site reactions. Its peptide nature allows for predictable metabolism and clearance, primarily through enzymatic degradation, minimizing concerns about long-term toxicity. Advances in formulation and peptide chemistry have further enhanced its stability, enabling convenient storage and clinical use. Overall, cetrorelix represents a clinically important GnRH antagonist, widely applied in reproductive medicine and hormone-sensitive disease management. Its design reflects a balance of receptor specificity, peptide stability, and clinical efficacy, demonstrating the potential of synthetic peptide analogues in targeted endocrine therapies. References Al-Inany HG, Youssef MA (2013) Gonadotropin-releasing hormone antagonists in assisted reproduction. Cochrane Database of Systematic Reviews 2 CD001750 DOI: 10.1002/14651858.CD001750.pub3 |
| Market Analysis Reports |