Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Sichuan Hongguang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.schgchem.com | |||
 | +86 (831) 339-2662 339-2277 339-2980 | |||
 | +86 (831) 330-0073 / 330-0016 | |||
 | hgwm@schgchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Shanghai Jinjinle Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jjlchem.com | |||
 | +86 (21) 6610-5180 5291-0829 5291-3935 | |||
 | +86 (21) 3201-0235 | |||
 | jjlchem@jjlchem.com | |||
| Chemical manufacturer | ||||
| AccuStandard Inc. | USA | |||
|---|---|---|---|---|
 | www.accustandard.com | |||
 | +1 (203) 786-5290 | |||
 | +1 (203) 786-5287 | |||
 | orders@accustandard.com | |||
| Chemical manufacturer since 1986 | ||||
| SelectLab Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.selectlab.de | |||
 | +49 (2383) 919-350 919-351 | |||
 | +49 (2383) 919-354 | |||
 | info@selectlab.de | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon nitrite |
|---|---|
| Name | 2,4-Dinitrotoluene |
| Synonyms | 1-Methyl-2,4-Dinitrobenzene; 2,4-Dinitrotoluol; 2,4-DNT |
| Molecular Structure | 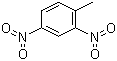 |
| Molecular Formula | C7H6N2O4 |
| Molecular Weight | 182.13 |
| CAS Registry Number | 121-14-2 |
| EC Number | 204-450-0 |
| SMILES | CC1=C(C=C(C=C1)[N+](=O)[O-])[N+](=O)[O-] |
| Water solubility | 0.3 g/L (20 °C) |
|---|---|
| Density | 1.4±0.1 g/cm3, Calc.* |
| Melting point | 67-70 °C (Expl.) |
| Index of Refraction | 1.598, Calc.* |
| Boiling Point | 299.9±20.0 °C (760 mmHg), Calc.*, 300 °C (Expl.) |
| Flash Point | 146.9±14.6 °C, Calc.*,155 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS06;GHS08;GHS09 Danger Details GHS06;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H350-H341-H361f-H331-H311-H301-H373-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P262-P264-P270-P271-P273-P280-P284-P301+P316-P302+P352-P304+P340-P316-P318-P319-P320-P321-P330-P361+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2038; UN 1600; UN 3454 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2,4-Dinitrotoluene is a chemical compound belonging to the class of nitrotoluenes, which are derivatives of toluene with two nitro groups (-NO2) attached to the benzene ring. The compound is one of several isomers of dinitrotoluene and has found applications in various industrial processes, including the synthesis of explosives, dyes, and pharmaceuticals. Its chemical structure, which consists of a toluene backbone with two nitro groups positioned at the 2 and 4 positions, gives it unique physical and chemical properties that make it a valuable intermediate in chemical synthesis. The discovery of 2,4-dinitrotoluene can be traced back to the late 19th century, when the nitration of toluene with a mixture of concentrated nitric and sulfuric acids was first explored. This reaction was of particular interest to chemists because it resulted in the formation of several isomers, each with different positions for the nitro groups on the aromatic ring. Among these isomers, 2,4-dinitrotoluene was identified as one of the most important and widely studied due to its high reactivity and versatile applications. One of the primary uses of 2,4-dinitrotoluene is in the synthesis of explosives, particularly in the production of TNT (trinitrotoluene). TNT is synthesized through a series of nitration reactions starting from dinitrotoluene, and the compound plays a crucial role in the manufacturing of this highly energetic material. In addition to its role in explosive production, 2,4-dinitrotoluene is used in the synthesis of various chemical intermediates, such as dyes, pigments, and pharmaceuticals. The nitro groups in 2,4-dinitrotoluene contribute to its chemical reactivity, allowing it to undergo a variety of reactions, including reduction and substitution, which are important in the preparation of these materials. In the field of pharmaceuticals, 2,4-dinitrotoluene has been explored for its potential use as a precursor for the synthesis of certain drug molecules. The compound's ability to undergo selective reductions and functionalizations makes it a useful building block for the creation of more complex therapeutic agents. For instance, derivatives of 2,4-dinitrotoluene have been investigated for their potential antibacterial and anticancer properties, although further research is needed to fully understand their biological activities and efficacy in medical applications. The compound's use in chemical synthesis extends beyond explosives and pharmaceuticals. In the textile industry, 2,4-dinitrotoluene is utilized in the production of various dyes and pigments. The nitro groups on the aromatic ring contribute to the compound's ability to absorb light at specific wavelengths, making it a valuable intermediate in the creation of colored materials. The compound's chemical stability and ability to undergo further transformations make it a versatile reagent in the preparation of different types of synthetic dyes. Despite its usefulness in a variety of applications, 2,4-dinitrotoluene presents several environmental and health concerns. As a highly toxic and potentially carcinogenic compound, it poses risks to human health and the environment. Workers involved in the synthesis or handling of 2,4-dinitrotoluene must take precautions to avoid inhalation or skin contact, and proper disposal methods must be employed to prevent contamination of water and soil. Due to its persistence in the environment, there are ongoing efforts to develop safer alternatives to 2,4-dinitrotoluene in certain applications, particularly in the production of explosives and dyes. In conclusion, 2,4-dinitrotoluene is a chemical compound with significant industrial importance, particularly in the synthesis of explosives, dyes, and pharmaceuticals. Its discovery and development have paved the way for the production of essential materials, but its potential health and environmental risks highlight the need for careful handling and ongoing research into safer alternatives. References 1993. Cloning and characterization of Pseudomonas sp. strain DNT genes for 2,4-dinitrotoluene degradation. Journal of Bacteriology. DOI: 10.1128/jb.175.6.1831-1837.1993 2005. Engineering Pseudomonas fluorescens for Biodegradation of 2,4-Dinitrotoluene. Applied and Environmental Microbiology. DOI: 10.1128/aem.71.12.8864-8872.2005 2018. Evolving metabolism of 2,4‐dinitrotoluene triggers SOS‐independent diversification of host cells. Environmental Microbiology. DOI: 10.1111/1462-2920.14459 |
| Market Analysis Reports |