Ammonium bromide, with the chemical formula NH₄Br, is an inorganic compound that exists as a white crystalline solid. It is commonly used in various applications in industries ranging from pharmaceuticals to photography, as well as in certain chemical syntheses. Ammonium bromide is typically produced by reacting ammonia gas with hydrogen bromide or a bromine-containing compound. Its discovery can be traced to the early studies of ammonium salts and halogen chemistry in the 19th century, and it has since become a valuable compound in a variety of fields due to its versatile properties.
The history of ammonium bromide dates back to the early 19th century when chemists began exploring the properties of ammonia and its interaction with halogens. While ammonium salts such as ammonium chloride were well-known and widely used by this time, ammonium bromide was not isolated until researchers began experimenting with the reaction between ammonia and hydrogen bromide. By the mid-1800s, the compound was recognized for its unique properties, which set it apart from other ammonium salts. Its ability to form solid crystals and its relatively simple synthesis method made it an attractive chemical for various industrial applications.
One of the primary applications of ammonium bromide is in the field of photography, particularly in the production of photographic films and plates. In traditional silver-based photography, ammonium bromide was used as a component in photographic emulsions. It serves as a source of bromide ions, which are essential for the formation of light-sensitive silver bromide (AgBr) crystals. These crystals are key to the photographic process, where they react to light exposure, allowing for the creation of images. Although modern digital photography has largely replaced traditional film, ammonium bromide remains a part of the historical development of photographic technology.
In the pharmaceutical industry, ammonium bromide has been historically used as a sedative and in the treatment of conditions such as epilepsy and anxiety. The compound was once a common ingredient in various medicinal formulations, where it was thought to have a calming effect on the nervous system. However, the use of ammonium bromide in medicine has declined in favor of more effective and safer treatments. Despite this, it remains a useful chemical in the production of certain pharmaceutical intermediates and in the synthesis of other bromine-containing compounds.
Ammonium bromide also has applications in the chemical industry, where it is used in organic synthesis reactions. It acts as a source of bromide ions, which are useful in halogenation reactions, a process where a bromine atom is introduced into an organic molecule. These reactions are often utilized to synthesize compounds with specific chemical properties, which can be used in further manufacturing processes. Ammonium bromide is also employed as a flame retardant in some polymer materials, where it helps reduce flammability by interfering with the combustion process.
In addition to its industrial uses, ammonium bromide is used in the oil and gas industry, particularly in drilling fluids. It is added to water-based drilling fluids to control the density and improve the rheological properties of the fluid, making it more effective in maintaining pressure during drilling operations. This application is crucial for ensuring the safety and efficiency of drilling activities, especially in deep wells.
While ammonium bromide has many industrial uses, its handling requires caution. The compound can be harmful if ingested or inhaled, and prolonged exposure to its dust or vapors can irritate the respiratory tract, eyes, and skin. Like many ammonium salts, ammonium bromide should be stored and handled with care, particularly in industrial settings, where it is often used in large quantities.
In conclusion, ammonium bromide is a versatile compound with a variety of applications in industries such as photography, pharmaceuticals, chemical synthesis, and oil and gas. Though its medical use has declined, it continues to be a valuable chemical in several industrial processes. Its discovery and continued use highlight the importance of bromine chemistry in both historical and modern industrial practices.
References
2001. Use of WO42-on Layered Double Hydroxides for Mild Oxidative Bromination and Bromide-Assisted Epoxidation with H2O2. Journal of the American Chemical Society.
DOI: 10.1021/ja015930c
2018. Cationic surfactants as antifungal agents. Applied Microbiology and Biotechnology.
DOI: 10.1007/s00253-018-9467-6
2019. Halloysite nanotubes loaded with alkyl trimethyl ammonium bromide as antibacterial agent for root canal sealers. Dental materials : official publication of the Academy of Dental Materials.
DOI: 10.1016/j.dental.2019.02.018
|






















































































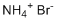

 GHS08;GHS07 Danger Details
GHS08;GHS07 Danger Details