Online Database of Chemicals from Around the World
| MolScanner | Singapore | |||
|---|---|---|---|---|
 | www.molscanner.com | |||
 | +86 18621675448 | |||
 | marketing@molscanner.com | |||
 | WhatsApp:9896 7603 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | dimethylethylpropynyloxypiperidinecarboxylate |
| Synonyms | tert-butyl 4-prop-2-ynoxypiperidine-1-carboxylate |
| Molecular Structure | 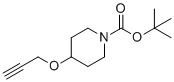 |
| Molecular Formula | C13H21NO3 |
| Molecular Weight | 239.31 |
| CAS Registry Number | 1219827-56-1 |
| SMILES | CC(C)(C)OC(=O)N1CCC(CC1)OCC#C |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 331.5±32.0 °C 760 mmHg (Calc.)* |
| Flash point | 154.3±25.1 °C (Calc.)* |
| Index of refraction | 1.49 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H312-H332 Details |
| Safety Statements | P261-P264-P270-P271-P280-P302+P352-P304+P340-P330-P363-P501 Details |
| SDS | Available |
|
1,1-Dimethylethyl 4-(2-propyn-1-yloxy)-1-piperidinecarboxylate is an organic compound characterized by a piperidine ring substituted at the nitrogen atom with a tert-butoxycarbonyl (Boc) protecting group and at the 4-position with a propargyloxy (2-propyn-1-yloxy) side chain. The structure is representative of synthetic intermediates used in medicinal and organic chemistry. The piperidine ring is a six-membered saturated heterocycle containing one nitrogen atom, commonly found in pharmaceutical compounds due to its favorable conformational flexibility and biological compatibility. In this molecule, the nitrogen atom is protected by a tert-butyl carbamate (Boc) group, which serves as a common amine protecting group. The Boc group is stable under basic conditions and is removed under acidic conditions, making it useful in multi-step organic synthesis. The presence of the 2-propyn-1-yloxy substituent introduces a terminal alkyne functional group to the molecule. Terminal alkynes are highly reactive and are frequently used in synthetic transformations such as the copper-catalyzed azide-alkyne cycloaddition (CuAAC), also known as "click chemistry." This feature makes the compound valuable for chemical conjugation, labeling, or the synthesis of more complex structures. 1,1-Dimethylethyl 4-(2-propyn-1-yloxy)-1-piperidinecarboxylate can be employed as a building block in drug discovery and development, particularly in the synthesis of piperidine-based pharmacophores. The Boc-protected amine allows for selective deprotection and further modification at the nitrogen atom, while the alkyne functionality offers a handle for attachment to various biological probes or reporter molecules. Although specific applications of this compound are typically proprietary or described in patents, compounds of this type have been widely used in the development of CNS-active drugs, enzyme inhibitors, and molecular probes. Its structure enables functional versatility, making it a useful intermediate in both combinatorial chemistry and targeted molecule design. References 2024. Pimonidazole-alkyne conjugate for sensitive detection of hypoxia by Cu-catalyzed click reaction. Analytical Sciences: The International Journal of the Japan Society for Analytical Chemistry, 40(5). DOI: 10.1007/s44211-024-00520-y |
| Market Analysis Reports |