Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| SINOJIE (HK) Limited | China | |||
|---|---|---|---|---|
 | www.sinojie.com | |||
 | +86 (21) 5230-3877 | |||
 | +86 (21) 5230-3799 | |||
 | sissizhou@sinojie.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Unibrom Corp. | China | |||
|---|---|---|---|---|
 | www.unibrom.com | |||
 | +86 (536) 829-1528 | |||
 | +86 (536) 829-1558 | |||
 | vvchem@gmail.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Dalian Synco Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.syncochemie.com | |||
 | +86 (411) 8363-5150 | |||
 | +86 (411) 8368-4420 | |||
 | daisy@syncochemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Yick-Vic Chemicals and Pharmaceuticals (HK) Ltd. | Hong Kong | |||
|---|---|---|---|---|
 | www.yickvic.com | |||
 | +852 2541-2772 | |||
 | +852 2542-3444 | |||
 | yickvic@hkstar.com | |||
| Chemical distributor since 1982 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Weifang Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qianjinchemical.com | |||
 | +86 (536) 517-0190 | |||
 | +86 (536) 889-7109 | |||
 | 1297429207@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Lyanchemicals Tech Co Ltd | China | |||
|---|---|---|---|---|
 | www.lyanchemicals.com | |||
 | +86 (21) 6406-5325 | |||
 | service@lyanchemicals.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Organica Feinchemie GmbH | Germany | |||
|---|---|---|---|---|
 | www.organica.de | |||
 | +49 (3494) 63-6215 | |||
 | +49 (3494) 63-6165 | |||
 | dmi@weylchem-organica.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Triethyl orthoformate |
| Synonyms | Ethyl orthoformate; Triethoxymethane |
| Molecular Structure | 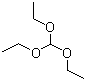 |
| Molecular Formula | C7H16O3 |
| Molecular Weight | 148.20 |
| CAS Registry Number | 122-51-0 |
| EC Number | 204-550-4 |
| SMILES | CCOC(OCC)OCC |
| Density | 0.891 |
|---|---|
| Melting point | -61 °C |
| Boiling point | 146 °C |
| Refractive index | 1.39-1.392 |
| Flash point | 30 °C |
| Water solubility | 1.35 g/L |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H319 Details | ||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P264+P265-P280-P303+P361+P353-P305+P351+P338-P337+P317-P370+P378-P403+P235-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2524 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Triethyl orthoformate, with the chemical formula C9H18O4, is an organosilicon compound commonly utilized as a reagent in organic synthesis. It is a colorless, volatile liquid that possesses a characteristic fruity odor. The compound was first synthesized in the early 20th century as part of a broader interest in formate esters and their potential applications in chemical reactions. The synthesis of triethyl orthoformate typically involves the reaction of ethanol with orthoformic acid. This process can also be accomplished by the esterification of formic acid with ethanol in the presence of an acid catalyst. The resulting product, triethyl orthoformate, is a member of the orthoformate family, which includes various esters that have found diverse applications in organic chemistry. One of the primary applications of triethyl orthoformate is in the synthesis of various organic compounds. It serves as a protective group for alcohols and amines, allowing chemists to selectively manipulate functional groups in complex molecules. This protective capability is particularly valuable in multi-step organic syntheses, where the protection and subsequent deprotection of functional groups can facilitate the formation of desired products. By using triethyl orthoformate, chemists can achieve greater control over the reactivity of alcohols, preventing unwanted side reactions during synthesis. In addition to its role as a protecting agent, triethyl orthoformate is commonly used as a reagent in the formation of acyl derivatives. It participates in various reactions, including acylation and esterification processes, enabling the introduction of acyl groups into molecules. This property makes triethyl orthoformate a useful building block in the synthesis of pharmaceuticals, agrochemicals, and other fine chemicals. Triethyl orthoformate is also employed in the preparation of polyurethanes and other polymeric materials. Its reactivity allows it to be incorporated into polymer matrices, enhancing the performance and properties of the resulting materials. In this context, triethyl orthoformate can improve the mechanical strength, chemical resistance, and thermal stability of polymers, making it an essential component in various industrial applications. Moreover, triethyl orthoformate has found applications in the field of analytical chemistry. It can be used as a derivatizing agent for the analysis of alcohols and amines, aiding in the identification and quantification of these compounds through techniques such as gas chromatography and mass spectrometry. This capability enhances the sensitivity and specificity of analytical methods, facilitating the detection of trace amounts of substances in complex mixtures. Despite its beneficial applications, handling triethyl orthoformate requires caution due to its flammable nature and potential health hazards. It can cause irritation to the skin, eyes, and respiratory tract upon exposure. Therefore, appropriate safety measures, including the use of personal protective equipment and proper ventilation, are essential when working with this compound in laboratory and industrial settings. In conclusion, triethyl orthoformate is a valuable reagent in organic synthesis, with applications ranging from protective group chemistry to polymer production and analytical methods. Its discovery and continued use underscore the importance of organosilicon compounds in the development of complex molecules and materials. Ongoing research and innovation in synthetic methodologies will likely further enhance the utility of triethyl orthoformate in various chemical applications. References 2011. Structure-Guided Evolution of Potent and Selective CHK1 Inhibitors through Scaffold Morphing. Journal of Medicinal Chemistry. DOI: 10.1021/jm2007326 2013. Quinazoline derivatives: synthesis and bioactivities. Chemistry Central Journal. DOI: 10.1186/1752-153x-7-95 2021. Strategies for synthesis of 1,2,4-triazole-containing scaffolds using 3-amino-1,2,4-triazole. Molecular Diversity. DOI: 10.1007/s11030-021-10197-4 |
| Market Analysis Reports |