Online Database of Chemicals from Around the World
| Topscience Co. Ltd. | China | |||
|---|---|---|---|---|
 | www.tsbiochem.com | |||
 | +86 (400) 820-0310 | |||
 | +86 (021) 3469-2979 | |||
 | sales@tsbiochem.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Hydride, nitride, azide |
|---|---|
| Name | TAMRA-PEG3-Azide |
| Synonyms | 5-[2-[2-[2-(2-azidoethoxy)ethoxy]ethoxy]ethylcarbamoyl]-2-[3-(dimethylamino)-6-dimethylazaniumylidenexanthen-9-yl]benzoate |
| Molecular Structure | 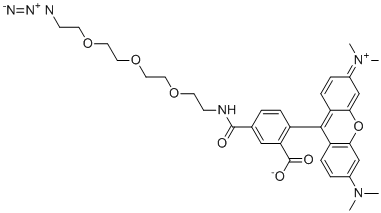 |
| Molecular Formula | C33H38N6O7 |
| Molecular Weight | 630.69 |
| CAS Registry Number | 1228100-59-1 |
| SMILES | CN(C)C1=CC2=C(C=C1)C(=C3C=CC(=[N+](C)C)C=C3O2)C4=C(C=C(C=C4)C(=O)NCCOCCOCCOCCN=[N+]=[N-])C(=O)[O-] |
| Hazard Symbols |  GHS07 Danger Details GHS07 Danger Details |
|---|---|
| Risk Statements | H228-H315-H319 Details |
| Safety Statements | P240-P210-P241-P264-P280-P302+P352-P370+P378-P337+P313-P305+P351+P338-P362+P364-P332+P313 Details |
| Transport Information | UN 1325 |
| SDS | Available |
|
TAMRA-PEG3-Azide is a synthetic chemical probe designed for bioorthogonal labeling and imaging applications, particularly in the context of click chemistry. The molecule combines three key structural elements: a tetramethylrhodamine (TAMRA) fluorophore, a triethylene glycol (PEG3) spacer, and a terminal azide group. Each component contributes distinct functional properties: TAMRA provides red fluorescence for detection and visualization; PEG3 improves solubility and reduces steric hindrance; and the azide moiety serves as a reactive handle for copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC), a widely used click chemistry reaction. The development of TAMRA-PEG3-Azide is rooted in the expanding field of bioorthogonal chemistry, which seeks to perform highly selective chemical transformations in biological environments without interfering with native biochemical processes. The azide group on TAMRA-PEG3-Azide is chemically inert to most cellular components, but reacts rapidly and selectively with alkynes under CuAAC conditions. This reaction forms a stable triazole linkage, enabling researchers to tag alkyne-containing biomolecules, such as modified nucleic acids, proteins, or glycans, with the fluorescent TAMRA moiety. TAMRA, short for 5(6)-carboxytetramethylrhodamine, is a well-established xanthene-based dye that exhibits strong absorption and emission in the orange-red region of the visible spectrum, typically with excitation around 546 nm and emission near 580–590 nm. Its brightness and photostability make it suitable for confocal microscopy, flow cytometry, and fluorescence resonance energy transfer (FRET) experiments. The carboxylic acid group on TAMRA allows for convenient conjugation to linkers such as PEG3, which in turn supports the introduction of functional groups like azides. The PEG3 spacer, composed of three ethylene glycol units, provides hydrophilicity and conformational flexibility. This segment acts as a linker to distance the bulky fluorophore from the site of azide-alkyne ligation, thereby minimizing steric hindrance and preserving biological function at the target site. PEG linkers are often employed in chemical biology for their ability to improve water solubility and reduce nonspecific interactions. TAMRA-PEG3-Azide has been extensively applied in chemical labeling strategies involving metabolic labeling or site-specific tagging. For example, cells can be metabolically engineered to incorporate alkyne-modified sugars or amino acids into their glycoconjugates or proteins. Upon treatment with TAMRA-PEG3-Azide and a copper catalyst, these biomolecules can be visualized by fluorescence microscopy, allowing researchers to track cellular processes such as glycosylation, protein synthesis, or cell proliferation. This labeling technique is both rapid and specific, making it an important tool in molecular and cell biology. In addition to imaging, TAMRA-PEG3-Azide can be used in pull-down assays or fluorescence-based quantification when combined with alkyne-functionalized probes or affinity handles. The high affinity and specificity of the CuAAC reaction ensure minimal background labeling, which is critical in experiments requiring high signal-to-noise ratios. Overall, TAMRA-PEG3-Azide exemplifies a multifunctional reagent that combines efficient fluorescence detection with versatile bioorthogonal reactivity. Its use has contributed to significant advances in labeling, tracking, and quantifying biomolecules in complex biological systems through non-invasive and highly selective chemical methodologies. References none |
| Market Analysis Reports |