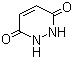
3,6-Dihydroxypyridazine is a heterocyclic compound belonging to the class of pyridazines, characterized by a six-membered aromatic ring containing two adjacent nitrogen atoms. Its molecular structure is defined by hydroxyl groups at the 3- and 6-positions of the pyridazine ring. This substitution pattern gives the compound distinct physicochemical and electronic properties, making it a subject of interest in both synthetic and medicinal chemistry.
The compound was first described in the literature through systematic studies of hydroxylated diazines, which aimed to understand the reactivity and potential biological activity of nitrogen-containing heterocycles. Its synthesis typically involves the hydroxylation of pyridazine derivatives under controlled conditions. One common route includes the use of dihydroxylating agents on precursor compounds such as pyridazine-3,6-dione or other suitably functionalized pyridazines. Alternatively, 3,6-dihydroxypyridazine can be obtained through diazotization and subsequent hydrolysis reactions starting from aminopyridazine derivatives.
Chemically, 3,6-dihydroxypyridazine exhibits tautomerism between keto and enol forms, although in aqueous or polar environments, the enol form with hydroxyl functionalities is generally favored. Its aromaticity and the presence of intramolecular hydrogen bonding between hydroxyl and nitrogen atoms influence its stability and reactivity. These structural features have been confirmed through spectroscopic methods, including nuclear magnetic resonance (NMR), ultraviolet-visible (UV-Vis) spectroscopy, and infrared (IR) spectroscopy.
In terms of application, 3,6-dihydroxypyridazine has been explored primarily as an intermediate in the synthesis of more complex molecules, especially in pharmaceutical and agrochemical research. It serves as a scaffold in the design of bioactive compounds due to the electron-rich nature of the pyridazine ring and the activating effect of the hydroxyl groups. Substituted pyridazines are known to exhibit a range of biological activities, including anti-inflammatory, antiviral, and anticancer properties. While 3,6-dihydroxypyridazine itself has not been approved as an active pharmaceutical ingredient, it is frequently utilized in synthetic schemes to build heterocyclic cores that are part of investigational or lead compounds.
Furthermore, the compound has utility in coordination chemistry as a potential bidentate ligand. The hydroxyl and nitrogen groups can coordinate with metal ions, making 3,6-dihydroxypyridazine suitable for the synthesis of metal complexes. These complexes are of interest for their potential catalytic activity and as models for metalloenzyme functions. Studies have demonstrated that such ligands can stabilize various oxidation states of metals and influence the geometry of metal coordination spheres.
In materials science, derivatives of 3,6-dihydroxypyridazine have been examined for their potential use in the development of organic electronic materials. The electron-donating capacity of the hydroxyl groups and the conjugated nature of the pyridazine ring support charge transport properties desirable in organic semiconductors. Although applications in this field are still in the early stages, structurally related compounds have shown promise in organic light-emitting diodes (OLEDs) and photovoltaic devices.
The compound also holds relevance in analytical chemistry, particularly in studies of hydrogen bonding, proton transfer, and solvation dynamics. Its simple structure yet well-defined donor and acceptor sites make it suitable for experimental and computational studies aimed at understanding intermolecular interactions in both gas and condensed phases.
In conclusion, 3,6-dihydroxypyridazine is a synthetically accessible and chemically versatile compound. Its role as a building block in organic synthesis, ligand in coordination chemistry, and subject in molecular interaction studies reflects its broad utility. While not extensively employed as an end-use product, its contribution to the development of functional molecules across multiple disciplines is well established in the literature.
References
2023. Maleic hydrazide prompting growth and delaying senescence of mother frond in S. Polyrriza 7498. Journal of Plant Physiology, 284.
DOI: 10.1016/j.jplph.2023.153966
2020. Maleic hydrazide elicits global transcriptomic changes in chemically topped tobacco to influence shoot bud development. Planta, 252(4).
DOI: 10.1007/s00425-020-03460-9
1949. Maleic Hydrazide, a Unique Growth Regulant. Science (New York, N.Y.), 109(2841).
DOI: 10.1126/science.109.2841.588-a
|


































































 GHS07;GHS08 Warning Details
GHS07;GHS08 Warning Details