Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Evans Chemetics LP | USA | |||
|---|---|---|---|---|
 | www.evans-chemetics.com | |||
 | +1 (201) 992-3100 | |||
 | +1 (201) 992-3101 | |||
 | j.westra@evans-chemetics.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Guangzhou Swan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.swanchemical.com | |||
 | +86 (20) 3104-4292 | |||
 | +86 (20) 3107-8112 | |||
 | helenlisa2003@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Qingdao ZKHT Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zkhtchem.com | |||
 | +86 (532) 8689-8076 +86 15153292017 | |||
 | +86 (532) 8689-8076 | |||
 | zkhtchem@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Other ester compounds |
|---|---|
| Name | Glycol dimercaptoacetate |
| Synonyms | Ethylene glycol bisthioglycolate; Ethylene glycol bis(mercaptoacetate); Mercaptoacetic acid 1,2-ethanediyl ester |
| Molecular Structure | 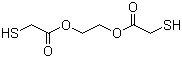 |
| Molecular Formula | C6H10O4S2 |
| Molecular Weight | 210.26 |
| CAS Registry Number | 123-81-9 |
| EC Number | 204-653-4 |
| SMILES | C(COC(=O)CS)OC(=O)CS |
| Density | 1.313 |
|---|---|
| Boiling point | 317 °C |
| Refractive index | 1.5175-1.5215 |
| Flash point | 167 °C |
| Water solubility | 20 g/L (20 °C) |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332-H302-H312-H315-H317-H319-H332-H335-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P272-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Glycol dimercaptoacetate, with the chemical formula C₅H₈O₄S₂, is a noteworthy compound recognized for its distinctive chemical properties and diverse applications. This organic compound, characterized by its glycol backbone and two mercaptoacetate groups, has been studied for its unique reactivity and utility in various industrial and research contexts. The discovery of glycol dimercaptoacetate emerged from the ongoing exploration of sulfur-containing organic compounds and their potential applications. Its synthesis involves the reaction of glycol with mercaptoacetic acid, leading to the formation of this compound with two thiol groups attached to a central glycol unit. The presence of mercapto groups imparts significant chemical reactivity, making glycol dimercaptoacetate a valuable compound in several fields. One of the primary applications of glycol dimercaptoacetate is in the field of polymer chemistry. The compound is used as a crosslinking agent in the synthesis of various polymer materials. Its ability to form disulfide bonds through thiol-ene chemistry makes it effective in creating crosslinked networks, which can enhance the mechanical properties and stability of polymers. This application is particularly relevant in the production of elastomers and hydrogels, where the crosslinking improves the material's elasticity and structural integrity. In addition to its use in polymer chemistry, glycol dimercaptoacetate serves as a key reagent in organic synthesis. Its thiol groups enable it to participate in various chemical reactions, including thiol-disulfide exchange reactions and Michael additions. These reactions are valuable for creating complex organic molecules and modifying existing compounds. As such, glycol dimercaptoacetate is utilized in the synthesis of pharmaceuticals, agrochemicals, and other specialty chemicals. The compound is also employed in the development of sensor materials. Its thiol groups can interact with metal ions and other reactive species, making it useful for detecting and quantifying specific substances. This property is leveraged in the creation of sensors for environmental monitoring and analytical chemistry. Despite its useful applications, glycol dimercaptoacetate must be handled with care due to the potential hazards associated with its mercapto groups. Proper safety protocols are necessary to minimize exposure and ensure safe usage in both industrial and laboratory settings. Overall, the discovery and application of glycol dimercaptoacetate underscore its significance in advancing polymer chemistry, organic synthesis, and sensor technology. Its unique chemical properties and versatility highlight its role in a range of industrial and research applications. References 1996. Some mechanistic insights in the behaviour of thiol containing antioxidant polymers in lignin oxidation processes. Research on Chemical Intermediates. DOI: 10.1163/156856796x00430 2015. Synthesis and characterization of novel antibacterial poly(imidosulfide)/Ag nanocomposite. Polymer Bulletin. DOI: 10.1007/s00289-015-1317-4 2024. Biodegradability of unsaturated poly(ester-thioether)s synthesized by thiol-yne reactions. Polymer Journal. DOI: 10.1038/s41428-024-00916-7 |
| Market Analysis Reports |