Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Suzhou Bichal Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bichal.com | |||
 | +86 (512) 6805-1130 | |||
 | +86 (512) 6805-2322 | |||
 | info@bichal.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Rochi Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rochipharma.com | |||
 | +86 (21) 3875-1876 +86 15000076078 | |||
 | +86 (21) 5027-5764 | |||
 | info@rochipharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Genriver Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.genriverpharm.com | |||
 | +86 13761582449 +86 13482015261 | |||
 | +86 (21) 3778-2903 | |||
 | info@genriverpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Changzhou Extraordinary Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.feifanchem.com | |||
 | +86 (519) 8211-1935 +86 15195055733 | |||
 | +86 (519) 8261-1250 | |||
 | info@feifanchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandong Chemed Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemedpharma.com | |||
 | +86 (531) 8880-3416 | |||
 | +86 (531) 8880-3416 | |||
 | sales@chemedpharma.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chsdpharma.com | |||
 | +86-531-58897029 | |||
 | ericqiao@jnchsd.com | |||
 | QQ Chat | |||
 | WeChat: 15550412551 | |||
 | WhatsApp:15550412551 | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Biochemical >> Inhibitor >> Protein tyrosine kinase |
|---|---|
| Name | Alectinib hydrochloride |
| Synonyms | 9-ethyl-6,6-dimethyl-8-(4-morpholin-4-ylpiperidin-1-yl)-11-oxo-5H-benzo[b]carbazole-3-carbonitrile;hydrochloride |
| Molecular Structure | 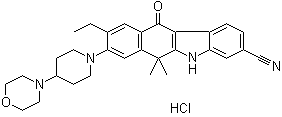 |
| Molecular Formula | C30H34N4O2.HCl |
| Molecular Weight | 519.08 |
| CAS Registry Number | 1256589-74-8 |
| EC Number | 814-550-1 |
| SMILES | CCC1=CC2=C(C=C1N3CCC(CC3)N4CCOCC4)C(C5=C(C2=O)C6=C(N5)C=C(C=C6)C#N)(C)C.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P280-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Alectinib hydrochloride is the hydrochloride salt form of alectinib, a highly selective, orally active small-molecule inhibitor of anaplastic lymphoma kinase (ALK). It is used in the treatment of ALK-positive non-small cell lung cancer (NSCLC), particularly in patients whose tumors harbor ALK gene rearrangements. The drug inhibits the kinase activity of ALK and several mutant forms associated with resistance to earlier ALK inhibitors, thereby preventing the phosphorylation and activation of downstream signaling pathways involved in cell proliferation and survival. The discovery of alectinib hydrochloride was led by Chugai Pharmaceutical Co., Ltd., a member of the Roche Group, as part of ongoing efforts to develop targeted therapies for genetically defined cancers. Alectinib was designed to overcome the limitations of first-generation ALK inhibitors, such as crizotinib, which often faced resistance due to secondary mutations in the ALK gene and limited central nervous system (CNS) penetration. Medicinal chemistry efforts focused on identifying a compound with strong potency against wild-type and mutant ALK variants, as well as favorable pharmacokinetic properties including brain penetration. Alectinib hydrochloride was approved in Japan in 2014 for ALK-positive NSCLC, followed by approvals in other countries. In the United States, it was granted accelerated approval by the U.S. Food and Drug Administration (FDA) in 2015 for patients with metastatic ALK-positive NSCLC who had progressed on or were intolerant to crizotinib. In 2017, it received full approval as a first-line treatment for ALK-positive metastatic NSCLC based on the results of randomized clinical trials demonstrating superior progression-free survival and CNS efficacy compared to crizotinib. The clinical development of alectinib hydrochloride was supported by multiple Phase I, II, and III trials. These studies showed that alectinib was effective in both systemic and CNS disease control, with a high overall response rate and durable clinical benefit. Its ability to penetrate the blood-brain barrier made it especially valuable for treating or preventing brain metastases, a common complication in ALK-positive NSCLC. Alectinib hydrochloride is administered orally and exhibits high selectivity for ALK, with minimal off-target activity. The drug is metabolized primarily by the cytochrome P450 enzyme CYP3A4, and its major active metabolite contributes to the overall pharmacologic effect. Alectinib has a relatively long half-life, supporting twice-daily dosing in clinical practice. In addition to its clinical efficacy, alectinib hydrochloride is generally well tolerated. Common side effects include fatigue, constipation, muscle pain, swelling, and anemia. Less frequent but potentially serious adverse effects include liver enzyme elevations, bradycardia, and interstitial lung disease. Regular monitoring of liver function and other laboratory parameters is recommended during treatment. The approval and widespread adoption of alectinib hydrochloride represent a significant advancement in the personalized treatment of lung cancer. By targeting the specific molecular abnormality driving tumor growth in a subset of NSCLC patients, the drug has improved outcomes and quality of life for individuals with ALK-rearranged disease. It continues to be studied in combination with other therapies and in earlier stages of disease to expand its clinical utility. In summary, alectinib hydrochloride is a next-generation ALK inhibitor developed for the treatment of ALK-positive NSCLC. Its discovery addressed critical needs in overcoming resistance and treating CNS involvement, and its clinical performance has established it as a cornerstone therapy in the precision medicine era for lung cancer. References 2014. Next-Generation Sequencing Reveals a Novel NSCLC ALK F1174V Mutation and Confirms ALK G1202R Mutation Confers High-Level Resistance to Alectinib (CH5424802/RO5424802) in ALK-Rearranged NSCLC Patients Who Progressed on Crizotinib. Journal of Thoracic Oncology, 9(4). DOI: 10.1097/jto.0000000000000094 2015. Severe acute interstitial lung disease in a patient with anaplastic lymphoma kinase rearrangement-positive non-small cell lung cancer treated with alectinib. Investigational New Drugs, 33(6). DOI: 10.1007/s10637-015-0284-9 2020. YAP1 mediates survival of ALK-rearranged lung cancer cells treated with alectinib via pro-apoptotic protein regulation. Nature Communications, 11(1). DOI: 10.1038/s41467-019-13771-5 |
| Market Analysis Reports |