Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Organic raw materials >> Organometallic salt |
|---|---|
| Name | Sodium 3-chloro-2-hydroxypropanesulfonate |
| Molecular Structure | 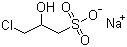 |
| Molecular Formula | C3H7ClO4S.Na |
| Molecular Weight | 196.58 |
| CAS Registry Number | 126-83-0 |
| EC Number | 204-807-0 |
| SMILES | C(C(CCl)O)S(=O)(=O)[O-].[Na+] |
| Melting point | 256 °C (Decomposes) (Expl.) |
|---|---|
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H318-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Sodium 3-chloro-2-hydroxypropanesulfonate is a water-soluble organosulfonate compound with the molecular formula C3H6ClNaO4S. It contains a three-carbon propane backbone substituted with a hydroxyl group at the second carbon, a chlorine atom at the third carbon, and a sulfonate group, existing as the sodium salt. This chemical is a crystalline solid, typically white or off-white, and is highly soluble in water due to its ionic sulfonate group. The compound is primarily synthesized through the nucleophilic addition of sodium bisulfite to epichlorohydrin. Epichlorohydrin, an epoxide with a three-carbon chain bearing a chlorine atom, undergoes ring-opening in the presence of bisulfite ions. The bisulfite ion attacks the epoxide ring, resulting in the formation of sodium 3-chloro-2-hydroxypropanesulfonate. This reaction is well documented in industrial chemistry and offers a high-yield, scalable route to this compound. The structural features of sodium 3-chloro-2-hydroxypropanesulfonate give it unique chemical and physical properties. The presence of the sulfonate group confers strong anionic character and excellent water solubility, which is valuable for its function in various applications. The hydroxyl group provides sites for potential hydrogen bonding and chemical reactivity, while the chlorine atom serves as a leaving group or site for further chemical modification. Industrially, this compound is widely used as a surfactant and dispersant. Its ability to solubilize in water and interact with both hydrophilic and hydrophobic substances makes it suitable for use in detergents, cleaning formulations, and emulsifiers. By incorporating this sulfonate moiety, formulations can achieve enhanced dispersion of particulate matter and improved stability against coagulation or precipitation. In polymer chemistry, sodium 3-chloro-2-hydroxypropanesulfonate is employed as a comonomer or additive to introduce sulfonate groups into polymer chains. This modification enhances the water solubility and ionic character of the polymers, which can improve properties such as adhesion, swelling behavior, and compatibility with aqueous systems. Such functionalized polymers find use in applications including coatings, adhesives, and water treatment membranes. Water treatment is another significant field for this compound. It acts as a dispersing agent, helping to prevent scale formation and deposition in industrial water systems by keeping minerals and particulates suspended and evenly distributed. This reduces fouling and extends the operational lifespan of equipment. Due to the chlorine substituent, sodium 3-chloro-2-hydroxypropanesulfonate can undergo further chemical transformations. For example, nucleophilic substitution reactions can replace the chlorine with other functional groups, allowing the synthesis of more complex sulfonated molecules tailored for specific industrial or chemical uses. The hydroxyl group similarly allows derivatization or polymer cross-linking. From a safety standpoint, this compound is moderately reactive due to its halogen content and should be handled with care. It can be an irritant to skin, eyes, and respiratory tract upon exposure. Therefore, standard industrial safety protocols including gloves, eye protection, and adequate ventilation are recommended during handling and processing. Storage should be in a cool, dry environment to maintain stability. In summary, sodium 3-chloro-2-hydroxypropanesulfonate is a versatile, water-soluble sulfonate salt synthesized via bisulfite addition to epichlorohydrin. Its chemical structure provides properties useful in detergents, dispersants, polymer modification, and water treatment. The compound’s reactive sites allow for further chemical modification, broadening its industrial applicability. Its handling requires standard safety precautions due to the presence of reactive chlorine. The compound’s established synthesis and multifaceted utility make it an important chemical intermediate and additive in several industrial sectors. References 2022. Synthesis and Performance Evaluation of Temperature and Salt-Resistant Foam Drainage Agent XY-1. Arabian Journal for Science and Engineering, 48(6). DOI: 10.1007/s13369-022-07531-9 2022. Synthesis and surface properties of sodium sulfonate amphoteric surfactants having different hydrophobic carbon chain length. Research on Chemical Intermediates, 48(5). DOI: 10.1007/s11164-022-04719-4 2017. Progress in the Synthesis of Zwitterionic Gemini Surfactants. Journal of Surfactants and Detergents, 20(6). DOI: 10.1007/s11743-017-2014-0 |
| Market Analysis Reports |