beta-Pinene is a bicyclic monoterpene widely recognized for its distinct aromatic properties and its role in various natural products. It has the molecular formula C₁₀H₁₈ and is one of the two isomers of pinene, the other being alpha-pinene. beta-Pinene is primarily found in the essential oils of various coniferous trees, particularly pine trees, and is also present in the oils of other plants such as rosemary and sage. The discovery of beta-pinene dates back to the 19th century when chemists began isolating and identifying the various components of essential oils. Its isolation was first achieved through the distillation of pine resin, leading to the recognition of its unique structure and potential applications.
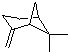
The structure of beta-pinene consists of a cyclobutane ring fused with a cyclohexene ring, creating a distinctive three-dimensional arrangement that contributes to its aromatic properties. The compound has a fresh, woody scent, making it popular in the fragrance and flavor industries. beta-Pinene is utilized in the production of perfumes, cosmetics, and various food flavorings due to its pleasant aroma. Its characteristic scent is often associated with fresh pine forests, making it a favored ingredient in products aimed at evoking a natural or outdoor experience.
In addition to its use in the fragrance industry, beta-pinene has garnered attention for its potential health benefits and therapeutic applications. Research has suggested that beta-pinene exhibits anti-inflammatory and antioxidant properties, making it a candidate for use in natural remedies and health supplements. Studies have indicated that beta-pinene may help alleviate symptoms of respiratory conditions and improve overall immune function. Its potential as a natural insect repellent has also been explored, providing an eco-friendly alternative to synthetic pesticides.
Moreover, beta-pinene has applications in the chemical industry, where it serves as a precursor for the synthesis of various compounds. It can be used to produce fragrances, flavors, and other chemical intermediates through various synthetic pathways. The ability to derive valuable chemical products from beta-pinene underscores its importance as a renewable resource, especially in the context of sustainable chemistry.
In conclusion, beta-pinene is a versatile and valuable chemical compound with a rich history of discovery and application. Its aromatic qualities make it a staple in the fragrance and flavor industries, while its potential therapeutic benefits and utility in chemical synthesis highlight its significance in both natural and industrial contexts.
References
2024. Chemical profiling and clustering of various dried cannabis flowers revealed by volatilomics and chemometric processing. Journal of Cannabis Research, 6.
DOI: 10.1186/s42238-024-00252-w
2024. Combating multidrug-resistant (MDR) Staphylococcus aureus infection using terpene and its derivative. World Journal of Microbiology and Biotechnology, 40.
DOI: 10.1007/s11274-024-04190-w
2011. Room temperature cationic polymerization of β-pinene using modified AlCl3 catalyst: toward sustainable plastics from renewable biomass resources. Green Chemistry, 13(9).
DOI: 10.1039/c1gc15593h
|




















































 GHS02;GHS07;GHS08;GHS09 Danger Details
GHS02;GHS07;GHS08;GHS09 Danger Details