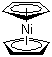
Nickelocene, or bis(cyclopentadienyl)nickel, is an organometallic compound with the chemical formula Ni(C5H5)2. It belongs to the metallocene family and is characterized by a nickel atom sandwiched between two cyclopentadienyl (C5H5) rings. Nickelocene is a dark green crystalline solid, which is air-sensitive and sublimates easily. Its structure and properties resemble those of other metallocenes like ferrocene, though it differs due to the unique electronic configuration of nickel.
The discovery of nickelocene dates back to the mid-20th century, when researchers explored the synthesis of transition metal complexes with cyclopentadienyl ligands. Nickelocene was first prepared by reacting nickel(II) chloride with sodium cyclopentadienide, forming the characteristic sandwich structure. Its synthesis helped expand the understanding of the bonding in transition metal complexes, particularly in terms of π-bonding interactions between the metal and the aromatic rings.
Nickelocene has found application in various fields due to its stability and electronic properties. It is used as a precursor in the preparation of nickel-based catalysts, which are essential in many industrial processes such as hydrogenation reactions and polymerizations. Nickelocene’s ability to serve as a source of nickel atoms in these reactions is critical, especially in the development of homogeneous catalysts.
In materials science, nickelocene has been employed in the chemical vapor deposition (CVD) process to create thin nickel films. These films are used in the production of electronics, particularly in applications that require precise deposition of nickel layers. The ability to control the deposition at a molecular level makes nickelocene a valuable material in the fabrication of nanostructures and advanced electronic devices.
Nickelocene is also studied in the context of organometallic chemistry and molecular electronics. Its electronic properties and magnetic behavior make it a subject of interest in the design of molecular magnets and conductive materials. Researchers continue to investigate its potential in creating novel materials with tunable electronic and magnetic properties, which could have implications for future technologies in data storage and quantum computing.
References
2007. Hawley's Condensed Chemical Dictionary, 15th Ed. New York: John Wiley & Sons.
2005. Toxicological profile for nickel. ATSDR - Agency for Toxic Substances and Disease Registry, U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). Available at: http://www.atsdr.cdc.gov/toxprofiles/tp15.html
1983. Activation of calcineurin by nickel ions. Biochemical and Biophysical Research Communications, 114(3), 955-961.
DOI: 10.1016/0006-2952(83)90660-3
|

































 GHS02;GHS07;GHS08 Danger Details
GHS02;GHS07;GHS08 Danger Details