Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Catalysts and additives >> Antioxidant |
|---|---|
| Name | 2,6-Di-tert-butyl-4-methylphenol |
| Synonyms | 2,6-Bis(1,1-dimethylethyl)-4-methylphenol; 2,6-Di-tert-butyl-p-cresol; BHT; Butylated hydroxytoluene; DBPC |
| Molecular Structure | 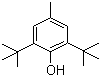 |
| Molecular Formula | C15H24O |
| Molecular Weight | 220.35 |
| CAS Registry Number | 128-37-0 |
| EC Number | 204-881-4 |
| SMILES | CC1=CC(=C(C(=C1)C(C)(C)C)O)C(C)(C)C |
| Density | 1.048 |
|---|---|
| Melting point | 69-71 °C |
| Boiling point | 265 °C |
| Flash point | 127 °C |
| Water solubility | insoluble |
| Hazard Symbols |  GHS09 Warning Details GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P273-P391-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2,6-Di-tert-butyl-4-methylphenol, commonly referred to as BHT (butylated hydroxytoluene), is a synthetic antioxidant widely used in various industrial and consumer applications. This compound was first synthesized in the 1930s as researchers sought to develop effective preservatives for food and other materials. Its discovery marked a significant advancement in the field of organic chemistry, particularly in the understanding of antioxidant properties and their applications in enhancing the stability of various substances. BHT is primarily recognized for its role as a food additive. It is used to prevent the oxidation of fats and oils, which can lead to rancidity and spoilage. By inhibiting oxidative degradation, BHT extends the shelf life of a wide range of food products, including snack foods, baked goods, and margarine. Its effectiveness in preserving flavor and color while maintaining nutritional quality makes it a valuable ingredient in the food industry. Furthermore, BHT is permitted for use in many countries, subject to regulatory limits, highlighting its importance in food preservation. In addition to its applications in food, BHT serves as a powerful antioxidant in various industrial products. It is commonly used in the formulation of cosmetics and personal care items, where it helps protect sensitive ingredients from oxidation, ensuring product stability and longevity. BHT is also found in lubricants, rubber, and plastics, where it prevents oxidative deterioration, improving the performance and durability of these materials. Its versatility as an antioxidant has made it a crucial component in numerous formulations across different industries. The mechanism by which BHT exerts its antioxidant effects involves the donation of hydrogen atoms to free radicals, thereby neutralizing them and preventing further oxidation of other compounds. This property not only enhances the stability of food and industrial products but also contributes to the overall safety and quality of these materials. By mitigating the formation of harmful oxidation products, BHT plays a vital role in maintaining the integrity of various substances throughout their shelf life. Despite its widespread use, the safety of BHT has been a subject of debate. Some studies have raised concerns about its potential health effects, including possible carcinogenicity and endocrine disruption. Regulatory agencies, including the Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA), have conducted extensive reviews of the available data and have established acceptable daily intake levels for BHT. These evaluations highlight the importance of monitoring its usage to ensure consumer safety while recognizing its benefits as an effective antioxidant. BHT can be synthesized through various methods, with the most common being the alkylation of 2,6-dimethylphenol with isobutylene in the presence of an acid catalyst. This synthesis route allows for the production of BHT on a commercial scale, making it readily available for use in food and industrial applications. In conclusion, 2,6-di-tert-butyl-4-methylphenol (BHT) is a significant chemical compound recognized for its antioxidant properties and diverse applications in the food and industrial sectors. Its discovery in the 1930s paved the way for advancements in food preservation and product formulation. While BHT remains an effective tool for enhancing the stability and safety of various products, ongoing research is essential to ensure its safe use in consumer applications. As industries continue to seek reliable antioxidants, BHT will likely remain a key player in the field of chemical preservatives. References 1979. Selective protective effect of butylated hydroxytoluene against 1,2-dimethylhydrazine carcinogenesis in BALB/c mice. Journal of the National Cancer Institute, 63(4). URL: https://pubmed.ncbi.nlm.nih.gov/480381 1984. Studies on the photoprotective mechanism of butylated hydroxytoluene. Photochemistry and Photobiology, 40(1). DOI: 10.1111/j.1751-1097.1984.tb04556.x 1994. Peroxide accumulation in detergents. Journal of Biochemical and Biophysical Methods, 29(1). DOI: 10.1016/0165-022x(94)90058-2 |
| Market Analysis Reports |