Online Database of Chemicals from Around the World
| Guizhou Jianye Yongrong Technology Co., Ltd. (Changzhou Huadong Chemical Research Institute) | China | |||
|---|---|---|---|---|
 | www.huadongchem.com | |||
 | +86 +13851563211 | |||
 | +86 (519) 8261-6386 | |||
 | sales@huadongchem.com | |||
 | WeChat: +13851563211 | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Surfactant >> Cationic surfactant >> Quaternary ammonium salt type |
|---|---|
| Name | N,N,N-Trimethyl-1-adamantanaminium chloride |
| Synonyms | 1-adamantyl(trimethyl)azanium chloride |
| Molecular Structure | 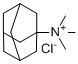 |
| Molecular Formula | C13H24ClN |
| Molecular Weight | 229.79 |
| CAS Registry Number | 128346-46-3 |
| EC Number | 801-489-0 |
| SMILES | C[N+](C)(C)C12CC3CC(C1)CC(C3)C2.[Cl-] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
|
N,N,N-Trimethyl-1-adamantanaminium chloride is a quaternary ammonium salt derived from adamantane, a rigid and highly symmetric hydrocarbon structure. It is formed by the methylation of the nitrogen atom in 1-adamantanamine, resulting in a positively charged nitrogen center and chloride as the counterion. This compound is typically used in research settings, especially in the context of its unique structural properties and potential applications in the fields of chemistry and material science. The compound has gained attention due to its structural similarity to other well-known quaternary ammonium compounds, which are often used in various chemical processes, including catalysis and organic synthesis. Its adamantane backbone, characterized by a rigid and non-planar structure, imparts interesting properties to the compound, which may influence its reactivity and interaction with other molecules. In terms of applications, N,N,N-Trimethyl-1-adamantanaminium chloride is primarily used in research related to organic chemistry, especially in the study of quaternary ammonium salts and their interactions with various substrates. It can be used as a phase transfer catalyst, a class of catalysts that facilitates the migration of reactants between different phases (e.g., between organic solvents and aqueous solutions). Phase transfer catalysts are important in synthetic chemistry, particularly in reactions where reactants need to be activated in different environments. Additionally, the compound has been studied for its potential use in materials science, especially in the development of new materials with specific properties. Its unique molecular structure may also make it a candidate for use in the development of new ionic liquids, which are solvents or electrolytes composed entirely of ions and have applications in a wide range of processes, including electrochemistry and catalysis. Despite its interesting properties, the broader commercial application of N,N,N-Trimethyl-1-adamantanaminium chloride is still largely limited to specialized academic and industrial research settings. Its primary use remains in studies focused on the behavior of quaternary ammonium compounds in various chemical and material systems. References 2020. Preparation method of adamantyl trimethyl ammonium hydroxide. CN-110699700-B. 2021. A kind of method and application of preparing SSZ-13 molecular sieve rich in aluminum pair. CN-112875720-B. |
| Market Analysis Reports |