Online Database of Chemicals from Around the World
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound |
|---|---|
| Name | 1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene |
| Synonyms | 3-[4-[1,2,2-tris(4-pyridin-3-ylphenyl)ethenyl]phenyl]pyridine |
| Molecular Structure | 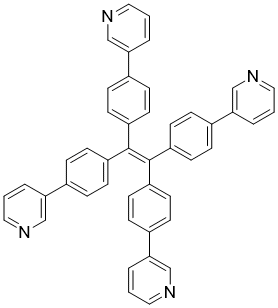 |
| Molecular Formula | C46H32N4 |
| Molecular Weight | 640.77 |
| CAS Registry Number | 1287777-24-5 |
| SMILES | C1=CC(=CN=C1)C2=CC=C(C=C2)C(=C(C3=CC=C(C=C3)C4=CN=CC=C4)C5=CC=C(C=C5)C6=CN=CC=C6)C7=CC=C(C=C7)C8=CN=CC=C8 |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.665, Calc.* |
| Boiling Point | 807.9±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 328.8±27.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene is an intriguing compound in the field of organic chemistry, known for its extended conjugated system and notable optical and electronic properties. This molecule features an ethene core with four pyridin-3-yl phenyl groups attached, creating a highly conjugated, planar structure. Its discovery and development have opened new avenues in material science, particularly in the realm of optoelectronics and sensing technologies. The compound was synthesized through advanced organic synthesis techniques, aiming to explore the effects of structural modifications on electronic and optical behavior. By replacing pyridin-4-yl groups with pyridin-3-yl groups, researchers sought to investigate how changes in substitution patterns impact the molecule's properties. The resulting compound, 1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene, exhibited significant alterations in its photophysical characteristics compared to its pyridin-4-yl analogs. One of the primary applications of 1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene is in the development of organic light-emitting diodes (OLEDs). The molecule's extended conjugated system facilitates efficient light emission when excited, making it a valuable component in OLED devices. Its ability to emit light in specific wavelengths allows for the creation of displays with vibrant colors and high brightness. Additionally, its stability and efficiency as an emissive material enhance the overall performance and lifespan of OLEDs. In organic photovoltaics (OPVs), 1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene serves as an important component due to its capacity for efficient charge transport and separation. The compound's conjugated structure helps in optimizing the absorption of sunlight and improving charge carrier mobility within the active layer of solar cells. By incorporating this compound, researchers aim to enhance the power conversion efficiency of OPVs and contribute to the advancement of sustainable energy technologies. The compound also finds application in sensor technologies. Its fluorescence properties allow it to act as a sensing material that can interact with various analytes. Changes in fluorescence upon interaction with specific substances enable the development of chemical sensors for environmental monitoring, medical diagnostics, and industrial applications. The versatility of 1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene in sensing applications stems from its ability to undergo changes in its optical properties in response to different chemical environments. Moreover, the molecule's unique structure and properties make it a subject of interest in supramolecular chemistry. The pyridine rings in 1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene can engage in non-covalent interactions, such as hydrogen bonding or coordination with metal ions, leading to the formation of complex supramolecular structures. These structures can exhibit novel properties and functions, which are valuable for designing new materials and catalysts. In summary, 1,1,2,2-Tetrakis(4-(pyridin-3-yl)phenyl)ethene represents a significant advancement in organic materials chemistry. Its discovery has led to various applications in optoelectronics, photovoltaics, sensing, and supramolecular chemistry, showcasing its versatility and impact on modern technology. References none |
| Market Analysis Reports |