Lithium hydroxide monohydrate, with the chemical formula LiOH·H₂O, is a white crystalline compound consisting of lithium hydroxide combined with one molecule of water. This hydrated form of lithium hydroxide is widely used in a variety of industrial and technological processes. Its discovery is closely linked to the exploration and refining of lithium compounds, which have gained prominence with the rising demand for advanced energy storage solutions, particularly lithium-ion batteries.
The isolation of lithium as an element occurred in 1817, when Johan August Arfvedson, a Swedish chemist, first identified it in the mineral petalite. However, lithium hydroxide monohydrate, as a specific compound, became more recognized as industrial chemists investigated various forms of lithium compounds for use in chemical reactions and applications. The production of lithium hydroxide monohydrate is typically achieved through the reaction of lithium carbonate (Li₂CO₃) with calcium hydroxide (Ca(OH)₂) in water, yielding LiOH·H₂O.
Structurally, lithium hydroxide monohydrate contains lithium cations (Li⁺) paired with hydroxide anions (OH⁻) and a single water molecule bound within the crystal lattice. This water molecule affects its physical properties, such as solubility and thermal stability. The compound is readily soluble in water, making it an effective reagent in both laboratory and industrial settings. As a strong base, it reacts with acids to form various lithium salts, expanding its utility in synthetic chemistry.
One of the most significant applications of lithium hydroxide monohydrate is in the production of lithium-ion batteries. Lithium hydroxide monohydrate is used to produce lithium compounds that form cathode materials, such as lithium cobalt oxide (LiCoO₂) and lithium nickel manganese cobalt oxide (NMC), which are integral to modern rechargeable batteries. These batteries are widely used in portable electronics, such as smartphones and laptops, and are now critical in electric vehicles (EVs), due to their high energy density and long cycle life. As the demand for EVs grows, so does the demand for high-purity lithium hydroxide monohydrate, as it enhances battery performance and longevity.
In addition to its role in battery manufacturing, lithium hydroxide monohydrate has significant industrial uses. One notable application is in the production of lubricating greases. Lithium hydroxide monohydrate reacts with fatty acids to form lithium soaps, which are key components in lithium-based greases. These greases exhibit excellent thermal stability, water resistance, and mechanical properties, making them ideal for use in automotive, aerospace, and industrial machinery applications.
Lithium hydroxide monohydrate also plays a critical role in air purification systems. Due to its ability to react with carbon dioxide (CO₂), it is used in closed environments, such as submarines and spacecraft, to remove CO₂ from the air. The reaction between lithium hydroxide monohydrate and CO₂ produces lithium carbonate (Li₂CO₃) and water, effectively scrubbing the air and maintaining breathable conditions in confined spaces. This application has been especially important in space exploration, where the maintenance of air quality is critical for astronaut safety.
Moreover, lithium hydroxide monohydrate is used in the ceramics industry, where it serves as a flux to lower the melting point of silica in glass production. This improves the strength and durability of glass products, making lithium hydroxide monohydrate an essential material in the manufacture of specialty glasses and ceramics.
With growing concerns about climate change, lithium hydroxide monohydrate has also been explored for its potential use in carbon capture technologies. Researchers are investigating its capacity to capture CO₂ from industrial emissions, offering a possible solution to reduce greenhouse gas concentrations in the atmosphere. This emerging application reflects the compound’s versatility and relevance in addressing global environmental challenges.
In summary, lithium hydroxide monohydrate is a crucial compound with diverse industrial and technological applications. Its importance in lithium-ion battery production, lubricants, air purification, and glass manufacturing underscores its versatility and the expanding role of lithium compounds in modern industries. As global demand for clean energy and advanced technologies continues to rise, lithium hydroxide monohydrate remains a key substance in driving innovation and sustainability.
References
1965. Colloidal properties and microstructure of lithium greases. Chemistry and Technology of Fuels and Oils, 1(8).
DOI: 10.1007/bf00730260
2024. Rationalize the High Performance of Lithium Sorbents Derived from Gibbsite Guided by Phase Chemistry. Journal of Sustainable Metallurgy, 10(2).
DOI: 10.1007/s40831-024-00839-w
2024. Lessons Learned from Developing Battery-Grade Lithium Projects. Proceedings of the 63rd Conference of Metallurgists, COM 2024.
DOI: 10.1007/978-3-031-67398-6_210
|



































































































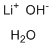


 GHS05;GHS06;GHS07 Danger Details
GHS05;GHS06;GHS07 Danger Details