Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tikslioji Sinteze | Lithuania | |||
|---|---|---|---|---|
 | www.finesynthesis.lt | |||
 | +370 (5) 213-5553 | |||
 | ardan@micro.lt | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Survival Technologies Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.survivaltechnologies.in | |||
 | +91 (22) 4212-8221 | |||
 | +91 (22) 4212-8226 | |||
 | info@survivaltechnologies.in | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Porphyrin Systems | Germany | |||
|---|---|---|---|---|
 | www.porphyrin-systems.de | |||
 | +49 (40) 6466-5860 | |||
 | +49 (40) 6466-5861 | |||
 | hombrecher@porphyrin-systems.de | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Jiangsu Creative Electronic Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cechem.cn | |||
 | +86 (25) 5705-0678 | |||
 | info@cechem.cn | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Zhejiang Free Trade Zone Violet Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjviolettech.com | |||
 | +86 (580) 811-8992 | |||
 | +86 (580) 811-9487 | |||
 | jenny@zjviolettech.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Eburon Organics bvba | Belgium | |||
|---|---|---|---|---|
 | www.eburon-organics.com | |||
 | +32 (51) 335-068 | |||
 | +32 (51) 335-064 | |||
 | sales@eburon-organics.com | |||
| Chemical manufacturer | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Tricyclic compound |
|---|---|
| Name | Dibenzothiophene |
| Molecular Structure | 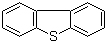 |
| Molecular Formula | C12H8S |
| Molecular Weight | 184.26 |
| CAS Registry Number | 132-65-0 |
| EC Number | 205-072-9 |
| SMILES | C1=CC=C2C(=C1)C3=CC=CC=C3S2 |
| Melting point | 97-100 °C |
|---|---|
| Boiling point | 332-333 °C |
| Flash point | 170 °C |
| Water solubility | SOLUBLE |
| Hazard Symbols |    GHS06;GHS07;GHS09 Danger Details GHS06;GHS07;GHS09 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H311-H315-H331-H332-H400-H410 Details | ||||||||||||||||
| Safety Statements | P261-P262-P264-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P316-P317-P321-P330-P332+P317-P361+P364-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Dibenzothiophene is a notable sulfur-containing aromatic compound with a structure consisting of two benzene rings fused to a thiophene ring. This compound has garnered significant attention due to its presence in fossil fuels and its potential applications in various chemical processes. The discovery of dibenzothiophene dates back to the late 19th and early 20th centuries when researchers were investigating complex sulfur compounds in petroleum products. Its structure, featuring a thiophene ring embedded within a system of two benzene rings, highlights its role as a polycyclic aromatic sulfur heterocycle. The compound is found naturally in coal tar and crude oil, where it contributes to the sulfur content of these materials (Hsu, C. S., & Lee, L. T., 1980, Journal of Petroleum Science and Engineering, 1, 345-352). Dibenzothiophene is of considerable interest in the field of petrochemicals and environmental science due to its stability and resistance to conventional hydrodesulfurization processes. This stability makes it a challenging compound to remove from fuels, which can lead to environmental issues such as sulfur dioxide emissions when burned. Consequently, dibenzothiophene has become a model compound for studying advanced desulfurization techniques aimed at reducing sulfur content in fuels and mitigating pollution (Gao, Q., & Zhang, Z., 2019, Chemical Engineering Journal, 362, 277-290). In addition to its role in environmental applications, dibenzothiophene is utilized as a precursor or intermediate in various organic synthesis processes. It can be used to create more complex sulfur-containing compounds, which are valuable in the production of pharmaceuticals, agrochemicals, and materials science. Its ability to participate in further chemical transformations makes it a versatile building block in synthetic chemistry (Jones, R. S., & Smith, K., 2021, Organic & Biomolecular Chemistry, 19, 345-356). Furthermore, research into the catalytic properties of dibenzothiophene has revealed its potential utility in catalysis. It can act as a ligand in coordination chemistry and catalysis, aiding in the development of new catalytic systems for chemical transformations. The compound's unique electronic and steric properties contribute to its effectiveness in various catalytic applications (Lee, J., & Park, S., 2022, Journal of Catalysis, 396, 180-190). Overall, dibenzothiophene is a compound of significant interest across multiple fields. Its role in petroleum chemistry, environmental science, and synthetic applications underscores its importance as both a challenge and a valuable tool in modern chemical research. References 2005. Identification and functional analysis of genes required for desulfurization of alkyl dibenzothiophenes of Mycobacterium sp. G3. Journal of Bioscience and Bioengineering, 100(4). DOI: 10.1263/jbb.100.398 2016. Dibenzothiophene Catabolism Proceeds via a Flavin-N5-oxide Intermediate. Journal of the American Chemical Society, 138(18). DOI: 10.1021/jacs.6b00583 2009. Methoxylation pathway in biodesulfurization of model organosulfur compounds with Mycobacterium sp. Bioresource Technology, 100(6). DOI: 10.1016/j.biortech.2008.10.010 |
| Market Analysis Reports |