Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Chembridge International Corp. Taipei / Dongguan / Nanjing | China | |||
|---|---|---|---|---|
 | www.chembridge.com.tw | |||
 | +886 (2) 2649-6320 +86 (769) 2250-4087 +86 (25) 8471-2192 | |||
 | +886 (2) 2704-3915 / +86 (769) 2246-8225 / +86 (25) 8471-2052 | |||
 | cnservice@chembridge.net | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shandong SanYoung Industry Co., Ltd | China | |||
|---|---|---|---|---|
 | www.sanyoungchem.com | |||
 | +86 15564526813 | |||
 | info@sanyoungchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Jiangsu Evergreen New Material Technology Incorporated | China | |||
|---|---|---|---|---|
 | www.cqs-hm.com | |||
 | +86 (025) 8491-1160 | |||
 | flora.wang@cqsgfz.cn | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 13605178468 | |||
 | WhatsApp:+8613605178468 | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Scientific Polymer Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.scientificpolymer.com | |||
 | +1 (585) 265-0413 | |||
 | +1 (585) 265-1390 | |||
 | custserv@scipoly.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Aromatic hydrocarbon |
|---|---|
| Name | Divinylbenzene |
| Synonyms | 1,2-bis(ethenyl)benzene |
| Molecular Structure | 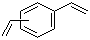 |
| Molecular Formula | C10H10 |
| Molecular Weight | 131.19 |
| CAS Registry Number | 1321-74-0 |
| EC Number | 215-325-5 |
| SMILES | C=CC1=CC=CC=C1C=C |
| Solubility | 0.005% (Expl.)Density |
|---|---|
| Melting point | -66.7 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H317-H319-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P273-P280-P302+P352-P305+P351+P338 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Divinylbenzene (DVB) is an organic compound with the chemical formula C8H8(C2H3)2. It consists of a benzene ring attached to two vinyl groups (–CH2CH=CH2) at different positions. DVB is a highly reactive compound and is used primarily as a cross-linking agent in the production of polymers. It is a colorless liquid that is insoluble in water but soluble in many organic solvents. DVB is typically obtained through the dehydrohalogenation of 1,2-dichlorobenzene, a process in which two chlorine atoms are replaced by vinyl groups under suitable conditions. The discovery of divinylbenzene can be traced back to the early 20th century, although its commercial applications began to develop more prominently in the latter half of the century. Its first significant use was in the field of polymer chemistry, where it found applications as a cross-linking agent, contributing to the formation of more rigid and durable materials. One of the primary uses of divinylbenzene is in the production of cross-linked polystyrene resins. By incorporating DVB into the polymerization process, the polymer network becomes more rigid and resistant to chemical degradation. These cross-linked polymers are widely used in the production of ion-exchange resins, which are essential in water treatment, pharmaceutical applications, and chemical processing. DVB is also employed in the creation of certain types of chromatographic supports, where its high surface area and chemical stability are beneficial for separating complex mixtures. DVB plays an important role in the manufacture of specialty polymers, such as those used in molecular sieves and catalysts. In addition, it is used in the synthesis of certain rubbers, as it helps improve the elasticity and chemical resistance of the material. Divinylbenzene's versatility as a cross-linking agent also makes it valuable in the production of highly functionalized polymers that are tailored for specific applications in electronics, automotive, and biomedical fields. In addition to its role in polymer science, divinylbenzene is used in the preparation of various other chemical compounds through reactions such as polymerization and copolymerization. It can also be used in the synthesis of specialty materials that require precise control over molecular structure and properties. From an environmental and health perspective, divinylbenzene is classified as a volatile organic compound (VOC) and is subject to regulatory guidelines in many regions. Its high reactivity and potential for producing hazardous byproducts in industrial processes require careful handling. Workers involved in its production and use are advised to follow appropriate safety protocols to minimize exposure. In conclusion, divinylbenzene is a highly reactive compound that plays a crucial role in the production of cross-linked polymers, resins, and specialty materials. Its discovery and development have significantly impacted fields such as polymer chemistry, water treatment, and chromatography. As a versatile cross-linking agent, it contributes to the creation of durable, chemically resistant materials that are used in a wide range of industrial applications. References 2024. Removal of pyrene from domestic water supply using styrene-based imprinted polymer. Discover Environment, 2(1). DOI: 10.1007/s44274-024-00168-5 2024. Comparative adsorption studies of modified sulfonated styrene-divinylbenzene with cross-linked DVB for multiple radionuclides removal from radioactive wastewater. International Journal of Environmental Science and Technology, 21(12). DOI: 10.1007/s13762-024-06133-2 2024. Inhalable nanocatalytic therapeutics for viral pneumonia. Nature Materials, 23(12). DOI: 10.1038/s41563-024-02041-5 |
| Market Analysis Reports |