Online Database of Chemicals from Around the World
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organotin |
|---|---|
| Name | Butylchlorodihydroxytin |
| Synonyms | Butylchlorodihydroxystannane |
| Molecular Structure | 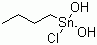 |
| Molecular Formula | C4H11ClO2Sn |
| Molecular Weight | 245.28 |
| CAS Registry Number | 13355-96-9 |
| EC Number | 236-406-1 |
| SMILES | CCCC[Sn](O)(O)Cl |
| Melting point | 150 °C (decomp.) |
|---|---|
| Boiling Point | 243.2±23.0 °C (760 mmHg), Calc.* |
| Flash Point | 100.9±22.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |     GHS05;GHS06;GHS08;GHS09 Danger Details GHS05;GHS06;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H312-H314-H317-H330-H341-H360-H370-H372-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P201-P202-P260-P264-P270-P271-P272-P273-P280-P284-P301+P310+P330-P301+P330+P331-P303+P361+P353-P304+P340+P310-P305+P351+P338+P310-P308+P311-P333+P313-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Butylchlorodihydroxytin, also known as butylchlorodihydroxytin(IV), is an organotin compound that has attracted attention due to its unique chemical properties and diverse applications, particularly in the field of materials science and as a biocide. The compound features a tin atom coordinated to a butyl group, two hydroxyl groups, and a chloride atom, which imparts specific characteristics that make it valuable in various industrial processes. The discovery of butylchlorodihydroxytin dates back to the 1970s when researchers were investigating organotin compounds for their potential applications in agriculture and industry. Organotin compounds were recognized for their antifungal and antibacterial properties, leading to their use as biocides and preservatives in a variety of products. The particular structure of butylchlorodihydroxytin allows it to exhibit effective antimicrobial activity while maintaining stability in different environmental conditions. One of the primary applications of butylchlorodihydroxytin is as a biocide in wood preservation. The compound is used to protect wood products from fungal decay and insect infestation. Its effectiveness against a wide range of microorganisms makes it a suitable choice for extending the lifespan of wood materials used in construction, furniture, and outdoor applications. The ability to prevent wood deterioration not only enhances the durability of products but also contributes to sustainable practices by reducing the need for frequent replacements. In addition to its use in wood preservation, butylchlorodihydroxytin has been explored for its applications in the production of polymers and coatings. The compound can be used as a stabilizer in the manufacture of polyvinyl chloride (PVC) and other polymeric materials. By incorporating butylchlorodihydroxytin into these materials, manufacturers can enhance their resistance to heat, light, and oxidation, thereby improving the overall performance and longevity of the products. However, the use of organotin compounds, including butylchlorodihydroxytin, has raised environmental and health concerns due to their toxicity. Studies have shown that some organotin compounds can be harmful to aquatic organisms and may bioaccumulate in the food chain. As a result, regulatory agencies have imposed restrictions on the use of certain organotin compounds in various applications. This has led to increased research into safer alternatives and the development of biocides with reduced environmental impact. Despite these challenges, butylchlorodihydroxytin continues to be of interest in the field of materials science. Researchers are investigating its potential in novel applications, such as antimicrobial coatings for medical devices and surfaces, where its effectiveness against a broad spectrum of pathogens can contribute to infection control. The ongoing exploration of its properties and potential uses in advanced materials demonstrates the compound's versatility and relevance in modern chemistry. The discovery of butylchlorodihydroxytin and its subsequent applications highlight the dual nature of organotin compounds as useful agents in industry while also raising awareness of their potential environmental implications. As the demand for sustainable and safe materials increases, the ongoing research into butylchlorodihydroxytin will play a crucial role in shaping its future applications and ensuring responsible use in various industries. References 1987. Organotin-induced hemolysis, shape transformation and intramembranous aggregates in human erythrocytes. Cell Biology and Toxicology, 3(2). DOI: 10.1007/bf00117823 2009. Synthesis, Characterization and Biological Studies of Oil Based Tin Polymer. Journal of Inorganic and Organometallic Polymers and Materials, 19(2). DOI: 10.1007/s10904-009-9288-2 2010. Impaired ergosterol biosynthesis mediated fungicidal activity of oil based tin polymer. Medicinal Chemistry Research, 20(4). DOI: 10.1007/s00044-010-9449-4 |
| Market Analysis Reports |