Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Cangzhou Goldlion Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.goldlionchem.com | |||
 | +86 13315721466 +86 (317) 530-4268 | |||
 | +86 (317) 530-4268 | |||
 | info@goldlionchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shijiazhuang Dowell Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dowechem.com | |||
 | +86 (311) 8980-5679 +86 13463963265 | |||
 | +86 (311) 8526-2695 | |||
 | sell@dowechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shijiazhuang Shengjia Chemical Co, Ltd. | China | |||
|---|---|---|---|---|
 | www.shengjiachem.com | |||
 | +86 (311) 8550-0777 +86 13930460855 | |||
 | +86 (311) 8550-0777 | |||
 | sales@shengjiachem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Jinhuan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinhchem.com | |||
 | +86 (21) 6142-0008 | |||
 | +86 (21) 5662-4506 | |||
 | chenhui79820@hotmail.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Novasyn Organics Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.novasynorganics.com | |||
 | +91 (406) 620-2233 633-9111 | |||
 | +91 (402) 781-1997 | |||
 | info@novasynorganics.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Porphyrin Systems | Germany | |||
|---|---|---|---|---|
 | www.porphyrin-systems.de | |||
 | +49 (40) 6466-5860 | |||
 | +49 (40) 6466-5861 | |||
 | hombrecher@porphyrin-systems.de | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Adesis, Inc. | USA | |||
|---|---|---|---|---|
 | www.adesisinc.com | |||
 | +1 (302) 323-4880 | |||
 | +1 (302) 323-4879 | |||
 | info@adesisinc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Eliv Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.elivpharma.com | |||
 | +86 (21) 5015-8063 | |||
 | +86 (21) 5015-8063 | |||
 | sales@elivpharma.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shijiazhuang C-element Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.c-elementbiotech.com | |||
 | +86 18033710157 | |||
 | Lisa@C-elementBiotech.com | |||
 | QQ Chat | |||
 | WeChat: 18033710157 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Activate Scientific GmbH | Germany | |||
|---|---|---|---|---|
 | www.activate-scientific.com | |||
 | +49 (8051) 965-1660 | |||
 | +49 (8051) 965-1661 | |||
 | rschmitt@activate-scientific.com | |||
| Chemical manufacturer | ||||
| Georganics Ltd. | Slovakia | |||
|---|---|---|---|---|
 | www.georganics.sk | |||
 | +421 (33) 640-3132 | |||
 | +421 (33) 640-3133 | |||
 | georganics@georganics.sk | |||
| Chemical manufacturer since 1998 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| True Pharmachem Inc. | USA | |||
|---|---|---|---|---|
 | www.truepharmachem.com | |||
 | +1 (217) 766-6932 | |||
 | +1 (973) 588-3470 | |||
 | sales@truepharmachem.com | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Other amino acid derivatives |
|---|---|
| Name | 2-Aminoisonicotinic acid |
| Synonyms | 2-Aminopyridine-4-carboxylic acid |
| Molecular Structure | 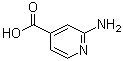 |
| Molecular Formula | C6H6N2O2 |
| Molecular Weight | 138.12 |
| CAS Registry Number | 13362-28-2 |
| EC Number | 681-100-7 |
| SMILES | C1=CN=C(C=C1C(=O)O)N |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 300-302 °C (Expl.) |
| Index of Refraction | 1.649, Calc.* |
| Boiling Point | 498.5±30.0 °C (760 mmHg), Calc.* |
| Flash Point | 255.3±24.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-Aminoisonicotinic acid is a pyridine derivative with the molecular formula C6H6N2O2. It consists of a pyridine ring substituted with an amino group at the 2-position and a carboxylic acid group at the 4-position. The compound is structurally classified under aminopyridinecarboxylic acids and is also known by its IUPAC name, 2-amino-4-pyridinecarboxylic acid. It appears as a solid and is typically soluble in polar solvents such as water and alcohols. The synthesis of 2-aminoisonicotinic acid has been documented in chemical literature and involves established methods starting from isonicotinic acid or its derivatives. One common synthetic approach includes nitration of isonicotinic acid to form the nitro derivative, followed by catalytic or chemical reduction to introduce the amino group at the desired position. This process yields 2-aminoisonicotinic acid with a high degree of regioselectivity. Other preparative routes include amination reactions under controlled conditions, using precursors such as halogenated pyridines or amide intermediates. The compound is primarily used as an intermediate in the preparation of various pharmaceuticals, agrochemicals, and heterocyclic compounds. Its functional groups, specifically the amino and carboxylic acid moieties, allow it to undergo a wide range of chemical transformations. These include amidation, esterification, acylation, and diazotization reactions. In pharmaceutical synthesis, 2-aminoisonicotinic acid serves as a building block for the development of molecules with antibacterial, antiviral, and anticancer properties. The aminopyridine framework is known for its utility in designing ligands that can interact with biological macromolecules. In coordination chemistry, 2-aminoisonicotinic acid acts as a bidentate ligand capable of coordinating to metal centers through both nitrogen and oxygen donor atoms. Metal complexes of 2-aminoisonicotinic acid have been synthesized and characterized, and they exhibit defined geometries and coordination behavior. These complexes are studied for their magnetic, catalytic, and electronic properties in materials science and inorganic chemistry. The compound has also been examined for its role in analytical chemistry as a reagent for detecting or quantifying certain metal ions. Its coordination capability makes it suitable for forming stable complexes with transition metals, which can be analyzed using spectroscopic or electrochemical techniques. This application has been reported in several studies focused on the development of analytical methods. Standard analytical techniques used to characterize 2-aminoisonicotinic acid include nuclear magnetic resonance (NMR) spectroscopy, infrared (IR) spectroscopy, ultraviolet-visible (UV-Vis) spectroscopy, and mass spectrometry (MS). The IR spectrum typically shows characteristic absorptions for N–H stretching vibrations of the amino group, C=O stretching of the carboxylic acid group, and C–N stretching. NMR spectra confirm the substitution pattern on the pyridine ring. In terms of physical properties, 2-aminoisonicotinic acid is stable under normal storage conditions and should be kept in a cool, dry place, protected from light and moisture. It is considered to have low acute toxicity but should be handled with care in accordance with standard laboratory safety procedures, including the use of gloves, protective clothing, and eye protection. Due to its established synthetic accessibility, functional versatility, and well-documented reactivity, 2-aminoisonicotinic acid continues to be an important compound in synthetic organic and inorganic chemistry. It is widely referenced in peer-reviewed chemical literature and remains a useful intermediate for academic and industrial research applications. References 2015. Microwave-Assisted Synthesis of 2,2'-Azopyridine-Labeled Amines, Amino Acids, and Peptides. Synthesis, 47(24). DOI: 10.1055/s-0035-1560523 2014. Synthesis and pharmacological evaluation of 6-aminonicotinic acid analogues as novel GABA(A) receptor agonists. European Journal of Medicinal Chemistry, 84. DOI: 10.1016/j.ejmech.2014.07.039 2010. Inhibition of chorismate-utilising enzymes by 2-amino-4-carboxypyridine and 4-carboxypyridone and 5-carboxypyridone analogues. Organic & Biomolecular Chemistry, 8(15). DOI: 10.1039/c004062b |
| Market Analysis Reports |