Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hengshui Haoye Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemcoms.com | |||
 | +86 (318) 210-2300 +86 18632882519 | |||
 | +86 (318) 210-3390 | |||
 | firmcn@gmail.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Xingtai City Meishen Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.mgochem.com | |||
 | +86 (319) 392-8532 | |||
 | +86 (319) 392-8502 | |||
 | sales9@mgochem.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Carbides and carbonates |
|---|---|
| Name | Magnesium carbonate |
| Synonyms | magnesite |
| Molecular Structure | 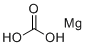 |
| Molecular Formula | CH2O3.Mg |
| Molecular Weight | 86.33 |
| CAS Registry Number | 13717-00-5 |
| EC Number | 604-004-9 |
| SMILES | [Mg].O=C(O)O |
| Density | 3.001 g/cm3 |
|---|---|
| Melting point | 2200 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Magnesium carbonate, with the chemical formula MgCO₃, is a versatile inorganic compound known for its diverse applications in various fields. This white, odorless powder is found naturally in the form of minerals such as magnesite and is commercially produced through various chemical processes. The discovery of magnesium carbonate dates back to the early 19th century, when it was first identified in mineral samples. Its natural occurrence in the form of magnesite was noted by scientists who were exploring mineral deposits. The synthesis of magnesium carbonate for industrial use began with the development of methods to obtain it from seawater or brine solutions, leveraging its solubility properties. In industrial applications, magnesium carbonate is widely used as a refractory material due to its high melting point and stability. It is employed in the production of magnesia bricks and other high-temperature ceramics, which are essential in the steel and glass industries. These materials provide thermal insulation and protect against corrosion in furnaces and kilns. Another significant application of magnesium carbonate is in the manufacturing of agricultural products. It is used as a soil conditioner and a source of magnesium in fertilizers. Magnesium is an essential nutrient for plant growth, and magnesium carbonate helps to correct magnesium deficiencies in soils, improving crop yields and quality. In the pharmaceutical industry, magnesium carbonate serves as an antacid. It neutralizes excess stomach acid, providing relief from indigestion and heartburn. Additionally, it is used as a dietary supplement to provide magnesium, which is crucial for various bodily functions, including nerve and muscle function, as well as bone health. Magnesium carbonate is also utilized in the production of personal care products, such as cosmetics and toothpaste. It acts as a bulking agent and a mild abrasive, contributing to the texture and effectiveness of these products. In sports, it is used by athletes and climbers as chalk to improve grip by reducing moisture on the hands. The compound's versatility extends to environmental applications as well. Magnesium carbonate can be employed in the treatment of wastewater and flue gases, where it helps in neutralizing acidic components and removing pollutants. Overall, magnesium carbonate is a valuable compound with a broad range of applications across different industries. Its discovery and subsequent uses highlight its importance in industrial processes, agriculture, pharmaceuticals, personal care, and environmental management. References 2017. Utilization of waste bittern from saltern as a source for magnesium and an absorbent for carbon dioxide capture. Environmental Science and Pollution Research International, 24(29). DOI: 10.1007/s11356-017-9913-5 1993. Successful Control of Hyperparathyroidism in Patients on Continuous Ambulatory Peritoneal Dialysis Using Magnesium Carbonate and Calcium Carbonate as Phosphate Binders. Nephron, 64(4). DOI: 10.1159/000187238 2024. Structural, electrical, and multiferroic investigations of MgBiFeTiO6 double perovskite for possible optoelectronic applications. Journal of Materials Science: Materials in Electronics, 35(32). DOI: 10.1007/s10854-024-13867-9 |
| Market Analysis Reports |