Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Manus Aktteva | India | |||
|---|---|---|---|---|
 | www.manusaktteva.in | |||
 | +91 (79) 6512-3395 | |||
 | +91 (79) 2646-3395 | |||
 | products@manusakttevabiopharma.in | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| VIVAN Life Sciences Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.vivanls.com | |||
 | +91 (22) 2544-4584 | |||
 | +91 (22) 2544-4588 | |||
 | vivan@vivanls.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Famo Bio-chemical Technology Company Ltd. | China | |||
|---|---|---|---|---|
 | www.famobiotech.com | |||
 | +86 (21) 6816-0314 | |||
 | +86 (21) 5816-6562 | |||
 | sales@famobiotech.com pharmabiotech@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Viwit Pharmaceuticals Limited | China | |||
|---|---|---|---|---|
 | www.viwit.com.cn | |||
 | +86 (21) 3777-5618 | |||
 | +86 (21) 3777-5619 | |||
 | service@viwit.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Beijing Leaderherb Bio-tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.china-api.cn | |||
 | +86 (10) 5128-8246 | |||
 | +86 (10) 5649-5849 | |||
 | 300abc@sina.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Unit Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharma-china.com | |||
 | +86 (27) 8787-3460 | |||
 | wendy@pharma-china.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Guangxi Shunjie Import & Export Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gxsjjck.com | |||
 | +86 (771) 8843-8884 | |||
 | catherine@gxsjjck.com | |||
| Chemical distributor since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Classification | API >> Digestive system medication >> Acid and gastric mucosal protective drugs |
|---|---|
| Name | (R)-Lansoprazole |
| Synonyms | 2-[[3-Methyl-4-(2,2,2-trifluoroethoxy)pyridin-2-yl]methylsulfinyl]-1H-benzoimidazole |
| Molecular Structure | 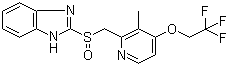 |
| Molecular Formula | C16H14F3N3O2S |
| Molecular Weight | 369.36 |
| CAS Registry Number | 138530-94-6 |
| EC Number | 688-930-9 |
| SMILES | CC1=C(C=CN=C1C[S@@](=O)C2=NC3=CC=CC=C3N2)OCC(F)(F)F |
| Density | 1.5±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.635, Calc.* |
| Boiling Point | 555.8±60.0 °C (760 mmHg), Calc.* |
| Flash Point | 289.9±32.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details |
|---|---|
| Risk Statements | H315-H317-H319-H335-H351-H373 Details |
| Safety Statements | P203-P260-P261-P264-P264+P265-P271-P272-P280-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
(R)-Lansoprazole is an enantiomer of lansoprazole, a widely used proton pump inhibitor (PPI) that reduces stomach acid production. Lansoprazole is commonly prescribed for conditions like gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. While both (R)-lansoprazole and its counterpart, (S)-lansoprazole, share similar pharmacological activities, (R)-lansoprazole is of particular interest due to its enhanced potency and pharmacokinetic profile in certain therapeutic applications. The discovery of lansoprazole itself is rooted in the quest for effective treatments for acid-related gastrointestinal disorders. In the late 1980s, researchers at the pharmaceutical company Takeda identified a need for compounds that could specifically inhibit the H+/K+-ATPase enzyme in the stomach lining, which is responsible for secreting gastric acid. Lansoprazole was developed as a member of the benzimidazole class, a group of compounds known for their ability to block this proton pump. The molecule exists as a racemic mixture of (R)- and (S)-enantiomers, with the (R)-enantiomer being the more biologically active form. (R)-Lansoprazole exerts its therapeutic effects by binding to and inhibiting the proton pump, thus preventing the secretion of gastric acid. This leads to a reduction in stomach acidity, which is beneficial in treating conditions such as GERD, where acid reflux can lead to symptoms like heartburn and damage to the esophagus. In addition to treating GERD, (R)-lansoprazole is also used in the management of peptic ulcers and in combination with antibiotics for the eradication of Helicobacter pylori, a bacterium linked to ulcer formation. One of the key advantages of (R)-lansoprazole over its racemic mixture is its improved pharmacokinetic profile. Studies have shown that (R)-lansoprazole is absorbed more efficiently in the body, leading to faster onset of action and potentially longer-lasting effects compared to the racemic compound. This makes (R)-lansoprazole a valuable option for patients who require more rapid and sustained acid suppression. Furthermore, (R)-lansoprazole may have a lower potential for side effects due to its more selective interaction with the proton pump enzyme. Beyond its use in acid-related gastrointestinal conditions, (R)-lansoprazole has also been explored for potential applications in other areas of medicine, including the prevention of nonsteroidal anti-inflammatory drug (NSAID)-induced ulcers. As the understanding of proton pump inhibitors continues to evolve, (R)-lansoprazole may play a larger role in managing a variety of gastrointestinal and related disorders. In conclusion, (R)-lansoprazole represents an important advancement in the treatment of acid-related gastrointestinal conditions. Its discovery as a more potent enantiomer of lansoprazole has led to its widespread clinical use, and ongoing research may uncover additional therapeutic benefits. The development of (R)-lansoprazole highlights the importance of understanding the distinct roles of drug enantiomers in optimizing patient outcomes. References 2002. Enantioselective disposition of lansoprazole in extensive and poor metabolizers of CYP2C19. Clinical pharmacology and therapeutics. DOI: 10.1067/mcp.2002.126176 2024. Potassium-competitive Acid Blockers: Current Clinical Use and Future Developments. Current Gastroenterology Reports. DOI: 10.1007/s11894-024-00939-3 2004. Pharmacokinetic differences between the enantiomers of lansoprazole and its metabolite, 5-hydroxylansoprazole, in relation to CYP2C19 genotypes. European Journal of Clinical Pharmacology. DOI: 10.1007/s00228-004-0809-1 |
| Market Analysis Reports |