Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Name | Methyl 4-oxobutyrate |
|---|---|
| Synonyms | 3-Formylpropionic acid methyl ester |
| Molecular Structure | 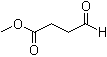 |
| Molecular Formula | C5H8O3 |
| Molecular Weight | 116.12 |
| CAS Registry Number | 13865-19-5 |
| EC Number | 237-611-9 |
| SMILES | COC(=O)CCC=O |
| Solubility | Soluble (46 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.037±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 167.3±23.0 °C (760 Torr), Calc.* |
| Refractive index | 1.4262 (589.3 nm 20 °C)** |
| Flash point | 60.3±22.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2013 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Methyl 4-oxobutyrate is synthesized by the condensation reaction of ethyl acetate with acetoacetic acid in the presence of an acidic catalyst. The compound produced by this reaction has the molecular formula C₅H₈O₃ and possesses a unique β-ketoester functional group. Methyl 4-oxobutyrate has a β-ketoester structure consisting of a methyl ester group attached to the β-carbon of a keto group (-CO-CH₂COOCH₃). This structure is both acidic and reactive, enabling it to participate in a variety of chemical reactions as both a nucleophile and an electrophile. Methyl 4-oxobutyrate is a colorless to pale yellow liquid at room temperature with a fruity aroma similar to that of apples. It is moderately soluble in water and highly soluble in organic solvents such as ethanol, acetone, and ether. The compound exhibits acidic properties due to the presence of keto and ester functional groups. It undergoes typical reactions associated with β-dicarbonyl compounds, including nucleophilic addition, condensation, and esterification reactions. Methyl 4-oxobutyrate is a key intermediate in organic synthesis, especially in the production of pharmaceuticals and fine chemicals. It undergoes condensation reactions with various nucleophiles to form β-diketones, which are valuable intermediates for the synthesis of vitamins, antibiotics, and other biologically active compounds. It participates in the synthesis of heterocyclic compounds such as pyrazoles, pyridines, and pyrroles. These heterocycles are fundamental structural motifs found in pharmaceuticals, agrochemicals, and materials science. Methyl 4-oxobutyrate is a precursor for the synthesis of acetoacetates, which are versatile intermediates for the preparation of β-keto acids and their derivatives. These compounds play a key role in medicinal chemistry and can be used to modify drug properties, improve bioavailability, and target specific biological pathways. It is also used in asymmetric synthesis to introduce chirality into molecules, enabling the production of enantiomerically pure compounds for use in pharmaceuticals and agrochemicals. Methyl 4-oxobutyrate contributes to fruity aroma and flavor profiles in food and beverage products. It is used as a flavoring agent in confectionery, beverages, and baked goods, enhancing their sensory appeal with its characteristic apple flavor. Methyl 4-oxobutanoate is a valuable tool for studying β-ketoester chemistry, reaction intermediates, and catalytic transformations. Its versatility and ease of handling make it an ideal substrate for exploring new synthetic approaches and reaction mechanisms. References 2016. Efficient synthesis of beraprost sodium. Chemical Research in Chinese Universities. DOI: 10.1007/s40242-016-6093-7 2022. Recent trends in Grubbs catalysis toward the synthesis of natural products: a review. Journal of the Iranian Chemical Society. DOI: 10.1007/s13738-021-02463-x 2015. Stereocontrolled Total Syntheses of Optically Active Furofuran Lignans. Synthesis. DOI: 10.1055/s-0034-1378812 |
| Market Analysis Reports |