Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Forever Synthesis Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.foreversyn.com | |||
 | +86 (551) 6288-8437 +86 18096409024 | |||
 | sales@foreversyn.com sales02@foreversyn.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | API >> Nervous system medication |
|---|---|
| Name | Tetrabenazine-d6 |
| Synonyms | rel-(3R,11bR)-1,3,4,6,7,11b-Hexahydro-9,10-di(methoxy-d3)-3-(2-methylpropyl)-2H-benzo[a]quinolizin-2-one; (RR,SS)-1,3,4,6,7,11b-Hexahydro-9,10-di(methoxy-d3)-3-(2-methylpropyl)-2H-benzo[a]quinolizin-2-one; Deutetrabenazine; SD 809 |
| Molecular Structure | 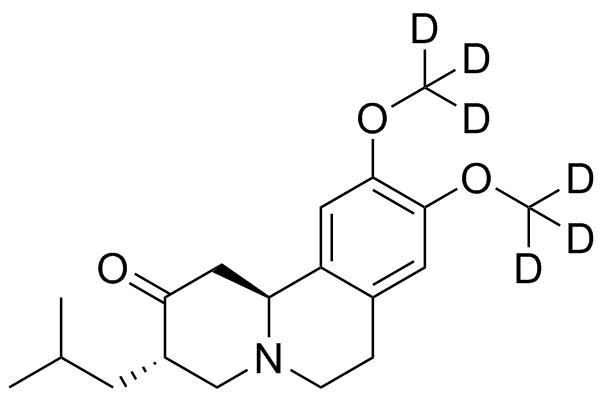 |
| Molecular Formula | C19H21D6NO3 |
| Molecular Weight | 323.46 |
| CAS Registry Number | 1392826-25-3 |
| EC Number | 813-698-4 |
| SMILES | [2H]C([2H])([2H])OC1=C(C=C2[C@@H]3CC(=O)[C@H](CN3CCC2=C1)CC(C)C)OC([2H])([2H])[2H] |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H336-H360-H361 Details | ||||||||
| Safety Statements | P203-P261-P271-P280-P304+P340-P318-P319-P403+P233-P405-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
|
Tetrabenazine-d6 is a deuterium-labeled isotopologue of tetrabenazine, in which six hydrogen atoms in the parent molecule are replaced with deuterium (2H). It is primarily used as an analytical reference standard in pharmacokinetic and bioanalytical studies rather than as a therapeutic drug itself. The compound belongs to the class of vesicular monoamine transporter 2 (VMAT2) inhibitors. The parent compound, tetrabenazine, was first synthesized in the mid-20th century and developed as a centrally acting agent that depletes monoamine neurotransmitters. It acts by reversibly inhibiting VMAT2, a transporter responsible for packaging monoamines such as dopamine, serotonin, and norepinephrine into synaptic vesicles. By blocking this transport, tetrabenazine reduces the amount of neurotransmitter available for release into the synaptic cleft, leading to decreased dopaminergic signaling in the central nervous system. Tetrabenazine-d6 retains the same core chemical structure as tetrabenazine but incorporates deuterium atoms at specific positions, typically at metabolically labile sites such as methyl groups. Deuterium is a stable isotope of hydrogen with one proton and one neutron, and its substitution can alter the rate of metabolic reactions involving C–H bond cleavage due to the kinetic isotope effect. However, in labeled standards such as tetrabenazine-d6, the primary purpose is not to modify pharmacological activity but to enable accurate analytical quantification. The development of deuterated analogues like tetrabenazine-d6 is closely associated with advances in mass spectrometry and pharmacokinetic research. These compounds are used as internal standards in liquid chromatography–mass spectrometry (LC-MS) assays. Because deuterated and non-deuterated compounds have nearly identical chemical and physical properties but different mass-to-charge ratios, tetrabenazine-d6 allows precise quantification of tetrabenazine and its metabolites in biological samples. The structure of tetrabenazine consists of a benzoquinolizine core with multiple methoxy and ester functional groups, which contribute to its lipophilicity and ability to cross the blood–brain barrier. The deuterium substitution in tetrabenazine-d6 does not significantly alter its chromatographic behavior, but it provides a measurable mass shift that improves analytical selectivity and accuracy in complex biological matrices such as plasma or tissue extracts. In pharmacological context, tetrabenazine is clinically used for the treatment of hyperkinetic movement disorders, including chorea associated with Huntington’s disease. Its mechanism of VMAT2 inhibition reduces excessive dopaminergic activity that contributes to involuntary movements. While tetrabenazine itself is therapeutically active, tetrabenazine-d6 is not used as a medication and is instead employed in research and diagnostic assay development. The synthesis of deuterated tetrabenazine analogues typically involves the incorporation of deuterium-labeled precursors during key steps of the synthetic route. This ensures that deuterium atoms are stably integrated into the final molecule without exchange under physiological or analytical conditions. The degree of labeling is controlled to achieve consistent isotopic enrichment for use as an internal standard. Overall, tetrabenazine-d6 is an isotopically labeled form of a VMAT2 inhibitor used primarily in analytical chemistry and pharmacokinetic studies. Its development reflects the broader application of stable isotope labeling in drug research, where structurally identical but mass-differentiated compounds enable precise measurement of drug concentrations and metabolic pathways in biological systems. References 2025. Exploring the Role of Deutetrabenazine in the Treatment of Chorea Linked with Huntington's Disease. Current Reviews in Clinical and Experimental Pharmacology. DOI: 10.2174/0127724328312991241001051813 2025. Safety and Efficacy of Deutetrabenazine at High versus Lower Daily Dosages in the ARC-HD Study to Treat Chorea in Huntington Disease. CNS Drugs. DOI: 10.1007/s40263-024-01139-3 2016. Effect of Deutetrabenazine on Chorea Among Patients With Huntington Disease: A Randomized Clinical Trial. JAMA. DOI: 10.1001/jama.2016.8655 |
| Market Analysis Reports |